Determine the phases present, the compositions of each phase, and the amount of each phase in mol%
Question:
Determine the phases present, the compositions of each phase, and the amount of each phase in mol% for the following NiO-MgO ceramics at 2400°C: (a) NiO- 30 mol% MgO;
(b) NiO-45 mol% MgO;
(c) NiO-60 mol% MgO; and
(d) NiO-85 mol% MgO.
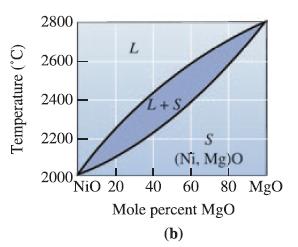
Transcribed Image Text:
Temperature (°C) 2800 2600 2400 2200 L 2000, L+S S (Ni. Mg)0 I NiO 20 40 60 80 MgO Mole percent MgO (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a The phases are NiO and MgO The ratio of NiO to M...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the phases that are present and the compositions for each phase in Cu-55 wt% Ag at 600C. Data Form Problem 11-23 Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or...
-
Determine the phases that are present and the compositions for each phase in Cu-85 wt% Ag at 800C. Temperature (C) 1200 1100 1000 900 800 700 600 500 400 300 200 0 10 20 30 40 50 60 70 80 90 100...
-
The unit cell of MgO is shown below. Does MgO have a structure like that of NaCl or ZnS? If the density of MgO is 3.58 g/cm3, estimate the radius (in centimeters) of the O2- anions and the Mg2+...
-
Calculate the following integral approximately by using the Simpson's method: 3.6 f. f(x) dx X 0.0 0.6 1.2 1.8 2.4 3.0 3.6 f(x) 0 0.13 0.05 0.6 0.43 0.74 0.93
-
Identify the advantages of automatic over policy adjustments to correct a trade disequilibrium.
-
Geodesic Domes, Inc., builds environmentally sensitive structures. The company's 2020 revenues totalled \(\$ 2,800\) million. At December 31,2020 , and 2019, the company had \$661 million and \(\$...
-
Refer to Exercise 10. The quality engineer suspects that the setting with a P-value of 0.03 may actually reduce the proportion of defective parts. What P-value would be needed to make this conclusion...
-
One end of a uniform 4.00-m-long rod of weight Fg is supported by a cable. The other end rests against the wall, where it is held by friction, as in Figure P12.23. The coefficient of static friction...
-
If a manager in a domiciliary care service is only providing personal care to one person at the time of the inspection, had not completed training recently to show they had the knowledge to...
-
The DellaVecchia Garden Center purchases and sells Christmas trees during the holiday season. It purchases the trees for $10 each and sells them for $20 each. Any trees not sold by Christmas day are...
-
What is the composition of the Monel alloy?
-
A Nb-W alloy held at 2800C is partly liquid and partly solid. (a) If possible, determine the composition of each phase in the alloy, and (b) If possible, determine the amount of each phase in the...
-
The graph shows the results of a study done by Guerin et al. (2013) and reported in the New England Journal of Medicine. Patients arriving at emergency rooms who were having trouble breathing were...
-
Assume that Fiskars Finland plans to invest in a new project that will annually generate revenues of \($2.3\) million. Cash expenses including both fixed and variable costs will be \($950,000,\) and...
-
Determine the IRR to the nearest percent on the following projects: a. An initial outlay of \($12,000\) resulting in a free cash flow of \($2,100\) at the end of year 1, \($5,210\) at the end of year...
-
Determine the IRR on the following projects: a. An initial outlay of \($12,000\) resulting in a free cash flow of \($2,041\) at the end of each year for the next 11 years b. An initial outlay of...
-
Determine the IRR on the following projects: a. An initial outlay of \($12,000\) resulting in a single free cash flow of \($17,231\) after 7 years b. An initial outlay of \($12,000\) resulting in a...
-
Holland at Home is considering introducing a variation of its current breakfast cereal, Zonnatura Regular Muesli Rich. The new cereal will be similar to the old with the exception that it will...
-
Cintas designs and manufactures uniforms for corporations throughout the United States and Canada. The company's stock is traded on the NASDAQ. Selected information from the company's financial...
-
What is an access control list?
-
a. Name each of the following alcohols. b. Name each of the following alcohols, including the stereochemistry if cistrans isomers are possible. HCH,CH,CH,CH,CH3 I CH3 T HCCH2CH 1 CH3 CH3
-
The two isomers having the formula C 2 H 6 O boil at -23 C and 78.5 C. Draw the structure of the isomer that boils at -23 C and of the isomer that boils at 78.5 C.
-
Consider the reaction of propane with chlorine (and appropriate catalyst). a. How many different monochloro products are possible? Name them. b. How many different dichloro products are possible?...
-
How do advanced indexing techniques, such as B-trees, hash indexing, and bitmap indexes, improve query performance in RDBMS, and what factors should be considered when choosing the appropriate...
-
Assume that CAPM holds. You are given the following information about stock X and the market portfolio, M: E(r) Riskless Asset (f) 0.05 (5%) 0.00 Stock X ? 0.40 Market Portfolio 0.10 0.20 (M) You are...
-
On January 1, 2024, Taco King leased retail space from Fogelman Properties. The 10-year finance lease requires quarterly variable lease payments equal to 3% of Taco King's sales revenue, with a...

Study smarter with the SolutionInn App


