A NiO-60 mol% MgO ceramic is allowed to solidify. Determine (a) The composition of the first solid
Question:
A NiO-60 mol% MgO ceramic is allowed to solidify. Determine
(a) The composition of the first solid to form, and
(b) The composition of the last liquid to solidify under equilibrium conditions.
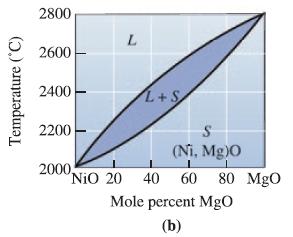
Transcribed Image Text:
Temperature (°C) 2800 2600 2400 2200 2000, L L+S S (Ni, Mg)0 NiO 20 40 60 80 MgO Mole percent MgO (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
a The first solid to form is composed of MgO oxygen and siliconoxygen eutectic it ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Under what conditions is a public company allowed to dismiss its independent audit firm? Under what conditions is an independent audit firm allowed to resign as the auditor of a public company? What...
-
Under what conditions is an accrual-basis taxpayer allowed to defer reporting amounts received in the advance of the delivery of goods?
-
A liquid solution contains 1 mol of CaCl2 and 25 mol of water. Using data from Pb. 12.37, determine the heat effect when an additional 1 mol of CaCl2 is dissolved isothermally in this solution. In...
-
How to drive target audience or people of Cineplex to movie theaters? Come up with a campaigns with big idea about Cineplex
-
Draw a figure similar to the right-hand panel of Figure 16.2 but with SX steeper (less elastic) than in Figure 16.2 and explain why S would be steeper (less elastic) than in Figure 16.1 if DX is...
-
Use the amortization table that you prepared for Standard Autoparts in exercise S7-9 to answer these questions about the company's long-term debt: 1. How much cash did Standard Autoparts borrow on...
-
The Bonferroni adjustment is made by multiplying the P-value by the number of___________________ . In Exercises 3 and 4, fill in each blank with the appropriate word or phrase.
-
Newland Company reported retained earnings at December 31, 2016, of $310,000. Newland had 200,000 shares of common stock outstanding at the beginning of 2017. The following transactions occurred...
-
Galtier Health Center is a nursing home in Minnesota, a state with the second largest Hmong population in the United States, behind only California (Pfeifer et al. 2013). The Hmong population...
-
Moss Co. issued $42,000,000 of five-year, 11% bonds, with interest payable semiannually, at a market (effective) interest rate of 9%. Determine the present value of the bonds payable using the...
-
What is macrosegregation? Is there a way to remove it without breaking up the cast structure?
-
What is homogenization? What type of segregation can it remove?
-
Suppose a risk-free asset has an expected return of 5 percent. By definition, its standard deviation is zero, and its correlation with any other asset is also zero. Using only asset A and the...
-
The period of a pendulum on the surface of the Earth is 1. It is then brought at a higher altitude. For it to maintain its period, should the length of the pendulum string be made longer or shorter?
-
A mass attached to a spring oscillates in simple harmonic motion along the \(x\)-axis. The limits of its motion are \(x=-5 \mathrm{~cm}\) and \(x=45 \mathrm{~cm}\) and it goes from one of these...
-
A certain metallic structure weighs \(2450 \mathrm{~N}\) on earth. The structure is sent to Mars, which has a diameter of \(6.78 \times 10^{6} \mathrm{~m}\) and mass of \(6.42 \times 10^{23}...
-
Two spherical celestial bodies, one of mass \(3.5 \times 10^{13} \mathrm{~kg}\) and the other of mass \(2.4 \times 10^{18} \mathrm{~kg}\), experience a gravitational force between them with a...
-
A battery and four identical light bulbs are arranged in the circuit of Figure P31.3. Rank the current magnitudes at the nine lettered locations from greatest to smallest. Data from Figure P31.3 90 e...
-
Computer Service and Repair was started five years ago by two college roommates. The company's comparative balance sheets and income statement are presented below, along with additional information....
-
The Higher the time period of the financial security the higher the. ............... risk. O a. Maturity O b. Default and Maturity Oc. Default O d. Liquidity
-
Consider the following four structures. a. Which of these compounds would have the same physical properties (melting point, boiling point, density, and so on)? b. Which of these compounds are trans...
-
Give two examples of saturated hydrocarbons. How many other atoms are bonded to each carbon in a saturated hydrocarbon?
-
Name the structural isomers in Exercise 15. Data in Exercise 15. Draw the five structural isomers of hexane (C 6 H 14 ).
-
In the modem Gregorian calendar (first adopted in 1582), an extra day (February 29) is added to the calendar to keep calendar years synchronized with astronomical years. Leap years occur according to...
-
Dandenong Health care is a provider of aged care services and currently employs 1000 employees with five centres in Victoria and seeks to expand its operations and open another office in Adelaide....
-
This Week's Detailed Case Study Information As the first light of day illuminates the BytePeak Software headquarters, a buzz of anticipation fills the air, much like the energy before the puck drop...

Study smarter with the SolutionInn App


