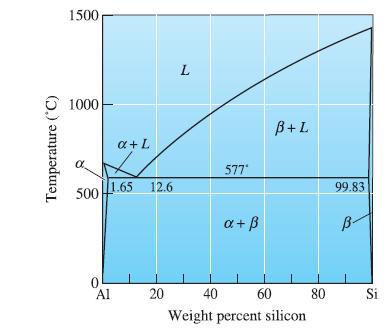
At the eutectic in the Al-Si phase diagram, what phase(s) is (are) present? Give a chemical analysis
Question:
At the eutectic in the Al-Si phase diagram, what phase(s) is (are) present? Give a chemical analysis of the phase(s).
Transcribed Image Text:
Temperature (°C) 1500 1000 500 1.65 0 a + L Al 12.6 20 L 577* a + ß B+L 40 60 Weight percent silicon 80 99.83 B- Si
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
ANSWER Step 1 Drawing of the aluminumsilico...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
The copper-silver phase diagram is shown in Figure 11-30. Copper has a higher melting point than silver. Figure 11-35 A phase diagram for elements A and B (for Problem 11-36). (a) Is copper element A...
-
A PT phase diagram for potassium is shown below. a. Which phase has the higher density, the fcc or the bcc phase? Explain your answer. b. Indicate the range of P and T in the phase diagram for which...
-
What is a phase diagram? Draw a generic phase diagram and label its important features.
-
For each of these utility functions, find the optimal consumption choices z and y for a consumer with budget w = 1, who faces prices pz = 0.05 and Py = 0.3. Are the goods substitutes or complements,...
-
(a) From Table 13.3, calculate the official settlements balance of the United States for each year from 1965 to 2011. (b) Why is this appropriate measure for the U.S. balance-of-payments position...
-
The prime numbers are those positive integers that are divisible by only 1 and themselves (for example, 2, 3, 5, 7, 11, 13, . . ). A celebrated theorem states that the sequence of prime numbers {p k...
-
What are examples of different types of systems?
-
Disclosure of Estimates Nancy Tercek, the financial vice president, and Margaret Lilly, the controller, of Romine Manufacturing Company are reviewing the financial ratios of the company for the years...
-
Blaze Corporation allocates overhead on the basis of DLH and the standard amount per allocation base is 3.20 DLH per unit. For March, the company planned production of 10,000 units (80% of its...
-
Describe the role of the police in contemporary society? 2. What does Alex Vitale argue? Do you agree with him? 3. Discuss the impact of the #Black Lives Matters protests. 4. How do you see the...
-
Consider an Al-4% Si alloy. Determine (a) If the alloy is hypoeutectic or hypereutectic; (b) The composition of the first solid to form during solidification; (c) The amounts and compositions of each...
-
An Al-Si alloy contains 85% and 15% at 500C. Determine the composition of the alloy. Is the alloy hypoeutectic or hypereutectic? Temperature (C) 1500 1000 500 1.65 12.6 0 a+L Al 20 L 577" a + B+L...
-
At December 31, 2025, Stevenson Company overstated ending inventory by $36,000. How does this error affect cost of goods sold and net income for 2025? a. Overstates cost of goods sold and understates...
-
Calculating ABC Unit Costs Perkins National Bank has collected the following information for four activities and two types of credit cards: Activity Driver Classic Gold Activity Rate ($) Processing...
-
Allied Merchandisers was organized on May 1. Macy Company is a major customer (buyer) of Allied (seller) products. May 3 Allied made its first and only purchase of inventory for the period on May 3...
-
Use the following selected account balances of Delray Manufacturing for the year ended December 31. Sales Raw materials inventory, beginning work in process inventory, beginning Finished goods...
-
You ask Alexa (a spherically symmetric speaker) to play your favorite Nickelback song. When you are standing 3.0 meters away, that sound reaches your ears at an intensity level of 50 decibels. Since...
-
Assuming no friction, if a falling ball arrives at the floor with kinetic energy of 350 J, what is its elastic potential energy when the ball comes to a complete stop?
-
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them: a. b. c. d. e. f. g. h. i. cis-2-butene and trans-2-butene j. CH3CH2CH2OH...
-
Assume today is the 21st of February. Using the information below, FT Extract, answer the following questions (parts i and ii). You work for a US company that is due to receive 250 million in June...
-
(a) Write the mass balance for CaCl 2 in water if the species are Ca 2+ and Cl - . (b) Write the mass balance if the species are Ca 2+ , Cl - , CaCl - , and CaOH + . (c) Write the charge balance for...
-
Write the charge and mass balances for dissolving CaF 2 in water if the reactions are CaF,(8) = Ca2+ + 2F Ca?+ + H,O = CAOH+ + H* Ca2+ + F = CaF+ CaF2(s) = CaF2(aq) F + H* = HF(aq) HF(aq) + F = HF,
-
Write charge and mass balances for aqueous Ca 3 (PO 4 ) 2 if the species are Ca 2+ , CaOH + , CaPO - 4 , PO 3- 4 , HPO 2 4 - , H 2 PO - 4 , and H 3 PO 4 .
-
A useful quantitative model for the grinding of lenses with known focal lengths is the lens-makers' equation: nlens environment 1) (1/4-1/2), where f is the focal length of the lens, the two index of...
-
Accelerating uniformly to overtake a slow-moving truck, a car moving initially at 24.4 m/s covers 67.2 m in 2.48 s. (a) What is the final speed of the car? m/s (b) What is the magnitude of the car's...
-
A student throws a set of keys vertically upward to her sorority sister, who is in a window 3.20 m above. The second student catches the keys 2.80 s later. (a) with what initial velocity were the...

Study smarter with the SolutionInn App


