Consider an Al-4% Si alloy. Determine (a) If the alloy is hypoeutectic or hypereutectic; (b) The composition
Question:
Consider an Al-4% Si alloy.
Determine
(a) If the alloy is hypoeutectic or hypereutectic;
(b) The composition of the first solid to form during solidification;
(c) The amounts and compositions of each phase at 578°C;
(d) The amounts and compositions of each phase at 576°C, the amounts and compositions of each microconstituent at 576°C; and
(e) The amounts and compositions of each phase at 25°C.
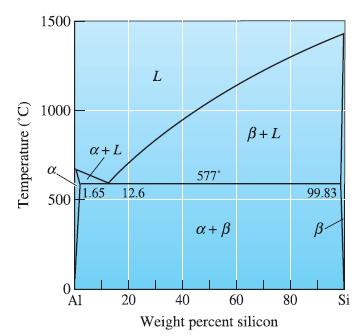
Transcribed Image Text:
1500 €1000 Temperature (C 500 1.65 12.6 0 a + L Al 20 L 577* a + ß B+L 40 60 Weight percent silicon 80 99.83 B- Si
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a The alloy is hypoeutectic The solidus temperature of the hypoeutectic ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or hypereutectic; (b) The composition of the first solid to form during solidification; (c) The amounts and compositions of each...
-
Consider an Al-4% Si alloy (Figure 11-19). Determine (a) If the alloy is hypoeutectic or hypereutectic; (b) The composition of the first solid to form during solidification; (c) The amounts and...
-
Consider an Al-12% Mg alloy (Figure 11-29). During solidification, determine (a) The composition of the first solid to form; (b) The liquidus temperature, solidus temperature, solvus temperature, and...
-
Use the data below to compute for the Gini coefficient. Round off the Gini coefficient to four (4) decimal places. INCOME CLASS (K) FIRST SECOND THIRD FOURTH FIFTH INCOME SHARE (YK) 0.02 0.12 0.28...
-
From the following figure, determine (a) The equilibrium exchange rate between the dollar and the pound sterling and the equilibrium quantity of pounds with supply curve S and S under a flexible...
-
Use the properties of infinite series to evaluate the following series. 3 (0.2)* + (0.8 k=0
-
What are the two primary types of systems associated with system, product, or service development?
-
The comparative balance sheets of Incloud Airlines show the following information for a recent year (amounts in thousands of US$): aCash was $378,511 at the beginning of the year and $418,819 at the...
-
Tariffs, import quotas, and export subsidies are the three sets of tools that governments commonly use to restrict international trade to protect domestic producers from foreign competition. (3.a)...
-
The firm Value Driver, Inc. has reported Revenues of $250 million in the year 2021. An equity analyst following the firm has projected the following information for the years 2022-2026: Expected...
-
A Pb-Sn alloy contains 23% primary a and 77% eutectic microconstituent immediately after the eutectic reaction has been completed. Determine the composition of the alloy.
-
At the eutectic in the Al-Si phase diagram, what phase(s) is (are) present? Give a chemical analysis of the phase(s). Temperature (C) 1500 1000 500 1.65 0 a + L Al 12.6 20 L 577* a + B+L 40 60...
-
Modify Exercise 4.15 to combine your code from the four separate triangles of asterisks such that all four patterns print side by side. Make clever use of nested for loops. Exercise 4.15 Write an...
-
Suppose you need to finance $ 2 0 , 0 0 0 for the purchase of a secondhand car. The interest rate on 4 - year auto loans is 1 2 % . What will your quarterly payment be ?
-
AJ is paid $14.52 per hour to work. He is paid time-and-a-half for all hours over 8 hours worked on a given day. Find his total earnings for the week if he works the following hours: Monday Tuesday...
-
Assume a restaurant chain is forced to pay damages to a person who suffered food poisoning after eating at the restaurant. What type of law is involved? Private law and civil law O Private law only...
-
Callable bond. Corso Books has just sold a callable bond. It is a thirty-year monthly bond with an annual coupon rate of 10% and $1,000 par value. The issuer, however, can call the bond starting at...
-
Bergo Bay's accounting system generated the following account balances on December 31. The company's manager knows something is wrong with this list of balances because it does not show any balance...
-
a. How could you determine by IR spectroscopy that the following reaction had occurred? b. After purifying the product, how could you determine that all the NH2 NH2 had been removed? CH3 CH NH2NH2...
-
Refer to the situation described inBE 18-13, but assume a 2-for-1 stock split instead of the 5% stock dividend. Prepare the journal entry to record the stock split if it is to be effected in the form...
-
Using activities, calculate the pH and concentration of H + in 0.050 M LiBr at 25C.
-
A 40.0-mL solution of 0.040 0 M Hg 2 (NO 3 ) 2 was titrated with 60.0 mL of 0.100 M KI to precipitate Hg 2 I 2 (K sp = 4.6 10 -29 ). (a) Show that 32.0 mL of KI are needed to reach the equivalence...
-
Write the mass balance for CaCl 2 in water if the species are Ca 2+ and Cl - . (b) Write the mass balance if the species are Ca 2+ , Cl - , CaCl - , and CaOH + . (c) Write the charge balance for part...
-
A steel cable that weighs 8 lb/ft is used to pull a 500 lb block of concrete from the ground to the top of a 120 ft tall building. Let x be the distance, in feet, from the block to the TOP of the...
-
A child is swinging a 330-g ball at the end of a 66.0-cm-long string in a vertical circle. The string can withstand a tension of 11.0 N before breaking. (a) What is the tension in the string when the...
-
Three metal fishing weights, each with a mass of 1.00102 g and at a temperature of 100.0C, are placed in 1.00102 g of water at 35.0C. The final temperature of the mixture is 45.0C. What is the...

Study smarter with the SolutionInn App


