Balance the following oxidationreduction reactions that occur in acidic solution using the half-reaction method. a. Cu(s) +
Question:
Balance the following oxidation–reduction reactions that occur in acidic solution using the half-reaction method.
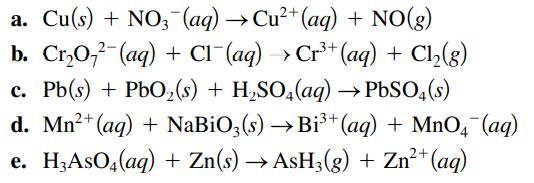
Transcribed Image Text:
a. Cu(s) + NO3(aq) → Cu²+ (aq) + NO(g) b. Cr₂O72 (aq) + Cl¯(aq) → Cr³+ (aq) + Cl₂(g) c. Pb(s) + PbO₂ (s) + H₂SO4(aq) → PbSO4(s) d. Mn²+ (aq) + NaBiO3(s) →Bi³+ (aq) + MnO4 (aq) e. H,AsO4(aq) + Zn(s) → AsH3(g) + Zn²+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
a Cus NO3aq Cuaq NOg Halfreaction Oxidation Cus Cuaq Reduction NO3aq N...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Balance each of the following oxidation reduction reactions by using the oxidation states method. a. C2H6(g) + O2(g) CO2(g) + H2O(g) b. Mg(s) + HCl(aq) Mg2+(aq) + Cl2(aq) + H2(g) c. Cu(s) + Ag+(aq)...
-
Balance these redox reactions that occur in aqueous solution. Use whatever water-derived species is necessary; there may be more than one correct balanced equation. a. CrO3 + Ni2+ ( Cr3+ + Ni3+ b....
-
Balance these redox reactions that occur in aqueous solution. Use whatever water-derived species is necessary; there may be more than one correct balanced equation. a. ClO + Ti2+ ( Ti4+ + Cl b. BrO3...
-
My division had another great year last year. We all worked hard, and the results were there. But again we got no reward for our hard work. It's very frustrating. - Division Manager, General Products...
-
What problem may exist in determining the amount realized for an investor who exchanges common stock of a publicly traded corporation for a used building? How is the problem likely to be resolved?
-
Travel and transfer to foreign locations are potential stressors for many people. Discuss your experiences with foreign travel or foreign assignments. Which parts of those experiences were the most...
-
Which intangible asset is recorded only as part of the acquisition of another company? a. Copyright b. Patent c. Franchise d. Goodwill
-
Upon the birth of his first child, Dick Jones decided to establish a savings account to partly pay for his son's education. He plans to deposit $20 per month in the account, beginning when the boy is...
-
55 Lavender Company's income under absorption costing was P3,600 lower than its income under variable costing. The company sold 10,000 units during the year, and its variable costs were P9 per unit,...
-
Find and analyze at least three sets of proposed principles for the governance of AI. What do the sets of principles have in common? How do they differ? How implementable are these principles?
-
Specify which of the following equations represent oxidation reduction reactions, and indicate the oxidizing agent, the reducing agent, the species being oxidized, and the species being reduced. a....
-
A 30.0-mL sample of an unknown strong base is neutralized after the addition of 12.0 mL of a 0.150 M HNO 3 solution. If the unknown base concentration is 0.0300 M, give some possible identities for...
-
The March 31, 2020, unadjusted trial balance for Silva Rentals after its first year of operations is shown below: Required 1. Enter the unadjusted trial balance onto a work sheet. 2. Using the...
-
How can identifying your job interests, abilities, and attributes help you develop an effective rsum?
-
What does it mean to view conflict as an opportunity?
-
In the working world, how might it be an advantage to be a high self-monitor? How might it be a disadvantage?
-
What is a shared story? What makes it useful for managing difficult conversations?
-
Why are exaggerating and taking either/or approaches counterproductive?
-
Tawnya Dobbs, the vice president of sales for Tropical Pools and Spas, wants the company's credit department to be less restrictive in granting credit. "How can we sell anything when you guys won't...
-
What kind of rays are X-rays?
-
A copper penny can be dissolved in nitric acid but not in hydro chloric acid. Using reduction potentials from the book, show why this is so. What are the products of the reaction? Newer pennies...
-
You want to plate out nickel metal from a nickel nitrate solution onto a piece of metal inserted into the solution. Should you use copper or zinc? Explain.
-
Galvanic cells harness spontaneous oxidationreduction reactions to produce work by producing a current. They do so by controlling the flow of electrons from the species oxidized to the species...
-
The structure of a Neural Network is given below. W W Y H X1 X2 H The input value X = X2 X-8 and the desired output value is Y The initial weight matrix for the first layer W: w = [WW] = [36] 4 (1)
-
Aysha's Cash Book showed a bank overdraft of Rs. 60,000 on 31" March, 2020. On Comparison of the two books, the following differences were noted down. a. A cheque for 3,000 deposited on 30th March,...
-
Fuzzy Monkey Technologies, Incorporated purchased as a long-term investment $140 million of 10% bonds, dated January 1, on January 1, 2024. Management intends to have the investment available for...

Study smarter with the SolutionInn App


