Carbon tetrachloride (CCl 4 ) and benzene (C 6 H 6 ) form ideal solutions. Consider an
Question:
Carbon tetrachloride (CCl4) and benzene (C6H6) form ideal solutions. Consider an equimolar solution of CCl4 and C6H6 at 25οC. The vapor above the solution is collected and condensed. Using the following data, determine the composition in mole fraction of the condensed vapor.
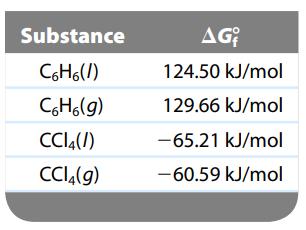
Transcribed Image Text:
Substance C6H6(/) C6H6(g) CC14(/) CC14(g) AG 124.50 kJ/mol 129.66 kJ/mol -65.21 kJ/mol -60.59 kJ/mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
Solution The composition in mole fraction of the conden...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
An equimolar solution of benzene and toluene is totally evaporated at a constant temperature of 90C. What are the pressures at the beginning and end of the vaporization process? Assume an ideal...
-
The diffusion coefficient of glucose in water at 25 C is 6.73 10 10 m 2 s 1 . Estimate the time required for a glucose molecule to undergo a root-mean-square displacement of 5.0mm.
-
Using the following data determine the equation of the regression model. How many independent variables are there? Comment on the meaning of these regression coefficients. Predictor .. Coefficient...
-
How could sales force automation affect salesperson productivity, marketing management, and competitive advantage?
-
How does each of the following events affect the risk of a liquidity trap? a. The central bank decides to push long-run inflation to zero. b. The neutral real interest rate rises. c. The government...
-
A uniform block of steel (SG =7.85) will float at a mercury-water interface as in the figure. What is the ratio of the distances a and b for this condition? Water block B Mercury: SG .3o
-
Why developing a theory of the case is important?
-
The Baby Store had a beginning inventory on January 1 of 200 full-size strollers at a cost of $110 per unit. During the year, the following purchases were made: ___________ Units _______ Unit Cost...
-
Dan owned a farm. Most of his products are being supplied to different restaurants. Products include cow's milk, chicken egg, meat (chicken, pork, and beef). On the side, they also sell carrots,...
-
Forth Company owned 85,000 of Brown Companys 100,000 shares of common stock until January 1, 20X2, at which time it sold 70,000 of the shares to a group of seven investors, each of whom purchased...
-
Tissue engineering involves the development of biological substitutes that restore or improve tissue function, Once manufactured, engineered organs can be implanted and grow within the patient,...
-
A 0.400-M solution of ammonia was titrated with hydrochloric acid to the equivalence point, where the total volume was 1.50 times the original volume. At what pH does the equivalence point occur?
-
Jones Inc. needs $100,000 to finance the purchase of new equipment. The finance manager is considering two options: 1. Borrowing the funds over a five-year term and paying interest at the rate of 6%...
-
The joint density function of two random variables \(X\) and \(Y\) is given by \[p_{X, Y}(x, y)= \begin{cases}\frac{x y}{9}, & 0 \leq x \leq 2,0 \leq y \leq 3 \\ 0, & \text { elsewhere }\end{cases}\]...
-
The standard normal variable, \(z\), corresponding to the normal variable \(x\), is defined as a. \(z=\frac{\bar{x}}{\sigma_{x}}\) b. \(z=\frac{x-\bar{x}}{\sigma_{x}}\) c. \(z=\frac{x}{\sigma_{x}}\)
-
The marginal density function of \(x\) can be determined form the bivariate density function \(p(x, y)\) as a. \(p(x)=\int_{-\infty}^{\infty} p(x, y) d y\) b. \(p(x)=\int_{-\infty}^{\infty} p(x, y) d...
-
The correlation coefficient of \(x\) and \(y\) is given by a. \(\sigma_{x y}\) b. \(\sigma_{x y} /\left(\sigma_{x} \sigma_{y} ight)\) c. \(\sigma_{x} \sigma_{y}\)
-
Compute the autocorrelation function of a periodic square wave with zero mean value and compare this result with that of a sinusoidal wave of the same period. Assume the amplitudes to be the same for...
-
A steel sheet has R values of 1.0, 1.5, and 2.0 for the 0, 45, and 90 directions to rolling, respectively. For a round blank 150 mm in diameter, estimate the smallest cup diameter to which it can be...
-
Explain the Hawthorne effect.
-
For the half-cell reaction AgBr(s) + e Ag(s) + Br (aq), E o = +0.0713 V. Using this result and G f (AgBr, s) = 96.9 kJ mol 1 , determine G o f (Br, aq).
-
For the half-cell reaction Hg 2 Cl 2 (s) + 2e 2Hg(l) + 2Cl (aq), E o = +0.26808 V. Using this result and G o f (Hg 2 Cl 2 , s) 210.7 kJ mol -1 , = determine G o f (Cl , aq).
-
Determine the half-cell reactions and the overall cell reaction, calculate the cell potential, and determine the equilibrium constant at 298.15K for the cell Is the cell reaction spontaneous as...
-
.There are 10 people waiting in line for a ride. Only 2 people can ride at a time. How many ways can 2 people go on the ride at once? You are going golfing. You have 3 drivers, 7 wedges, 6 irons, and...
-
The annual log returns on a portfolio is 15% and 10% separately over two years. Assume the initial investment on this portfolio is $10,000, what is the portfolio value at the end of the year two?
-
4 $81.47 5 $76.38 The following table summarizes prices of various default-free zero-coupon bonds (expressed as a percentage of the face value): Maturity (years) Price (per $100 face value) 1 $95.33...

Study smarter with the SolutionInn App


