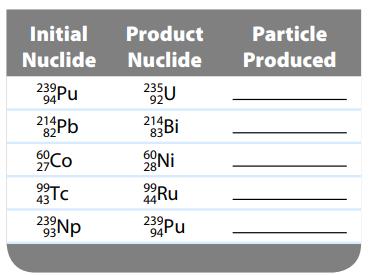
Complete the following table with the nuclear particle that is produced in each nuclear reaction. Initial Nuclide
Question:
Complete the following table with the nuclear particle that is produced in each nuclear reaction.
Transcribed Image Text:
Initial Nuclide 23⁹ Pu 94 214Pb 82 60, 27 99- 43 Tc 93Np Product Nuclide 2351 92 214p 60 Ni 28 44Ru 23⁹Pu 94 Particle Produced
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
ANSWER Types of radioactive particles The two protons and the two neutrons that make up an alpha par...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Complete the following table with either a yes or no regarding the attributes of a proprietorship, partnership and corporation. Proprietorship Attribute Present I. Business taxed. 2. Business entity...
-
Complete the following table with the correct formula related to various spread strategies. Bear Spread with Cals ith Puts with Calls Bull Spread Butterfly Spread Item Value at expiration Profit...
-
Complete the following table with the correct formula related to various spread strategies. Collar Strategies with Calls and Puts Straddle with Calls and Puts Item Value at expiration Profit Maximum...
-
A city starts a solid waste landfill in 2012. When the landfill was opened the city estimated that it would fill to capacity within 5 years and that the cost to cover the facility would be $1.5...
-
Consider the example of collateral in Section 7.5. The example assumes that collateral is $50. Determine the smallest level of collateral that causes the firm to choose the safe project. Assume the...
-
A free liquid jet, as in Fig P3.152, has constant ambient pressure and small losses; hence from Bernoullis equation z + V2/ (2g) is constant along the jet. For the fire nozzle in the figure, what are...
-
Compute the expected frequencies. Exercises 1015 refer to the following data: Anthropologists can estimate the birthrate of an ancient society by studying the age distribution of skeletons found in...
-
Variable costing versus absorption costing Lange Company incurred manufacturing overhead cost for the year as follows. The company produced 1,500 units and sold 1,000 of them at $180 per unit. Assume...
-
Harris Corp. has a corporate tax rate of 40%, has two sources of funds: long-term debt with a market value of $10,000,000 and an interest rate of 12%, and equity capital with a market value of...
-
Write pseudocode for each example (a through e) in Exercise 2, making sure your pseudocode is structured and accomplishes the same tasks as the flowchart segment. Data From Exercise 2 a. b. D Yes NO...
-
A certain radioactive nuclide has a half-life of 3.00 hours. a. Calculate the rate constant in s -1 for this nuclide. b. Calculate the decay rate in decays/s for 1.000 mole of this nuclide.
-
Strontium-90 and radon-222 both pose serious health risks. 90 Sr decays by -particle production and has a relatively long half-life (28.9 years). Radon-222 decays by a-particle production and has a...
-
Presented below is information related to Viel Company at December 31, 2012, the end of its first year of operations. Sales revenue $310,000 Cost of goods sold 140,000 Selling and administrative...
-
Solve Problem 11.6 by changing \(c\) to 2 . Data From Problem 11.6:- Find the free-vibration response of a viscously damped single-degree-of-freedom system with \(m=k=c=1\), using the central...
-
Why does dynamic balancing imply static balancing?
-
Integrate the differential equation \[-\frac{d^{2} x}{d t^{2}}+0.1 x=0 \quad \text { for } \quad 0 \leq t \leq 10\] using the backward difference formula with \(\Delta t=1\). Assume the initial...
-
Avoid excessive response at resonance a. Introduce damping b. Use vibration isolator c. Add vibration absorber d. Avoid resonance
-
Derive the formula for the fourth derivative, \(\left(d^{4} x ight) /\left(d t^{4} ight)\), according to the central difference method.
-
Describe the differences between compound, progressive, and transfer dies.
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
Propose structures for aromatic compounds that have the following 1 H NMR spectra: (a) C 8 H 9 Br IR: 820 cm ?1 (b) C 9 H 12 Br IR: 750 cm ?1 (b) C 11 H 16 Br IR: 820 cm ?1 TMS 10 8. 6. 0 ppm...
-
Propose a structure for a molecule C 14 H 12 that has the following 1 H NMR spectrum and has IR absorptions at 700, 740, and 890 cm ?1 : TMS 3 5 Chemical shift (8) 10 O ppm 8 7 Intensity-
-
Aromatic substitution reactions occur by addition of an electrophile such as Br+ to the aromatic ring to yield an allylic carbocation intermediate, followed by loss of fl. Show the structure of the...
-
What effect will a corporation's emphasis on environmental responsibility have on its financial performance in both the short term and long term? Does this contradict the notation of maximizing the...
-
Examine the emergence of technology and electronic health systems in health care since the passage of the Health Insurance Portability and Accountability Act (HIPAA). Provide an analysis of the...
-
Harris Fabrics computes its plantwide predetermined overhead rate annually on the basis of direct labor - hours. At the beginning of the year, it estimated that 70 , 0 0 0 direct labor - hours would...

Study smarter with the SolutionInn App


