Consider a galvanic cell based on the following theoretical half-reactions: What is the value of G
Question:
Consider a galvanic cell based on the following theoretical half-reactions:
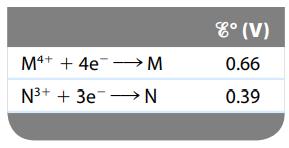
What is the value of ΔGο and K for this cell?
Transcribed Image Text:
M4+ + 4e→→→→→M N³+ + 3e N 8° (V) 0.66 0.39
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Its under conditions as well as the equilibrium constant K We know that this is a galvanic cell and ...View the full answer

Answered By

Dudhat Vaidehi
I tutored mostly elementary school students privately after school and during the summer. We met in their homes or at the public library. I charged an hourly fee, and I provided any necessary materials.
Having taught in special education in two local schools for many years meant that I had contact with a lot of parents of special needs students. I never had to advertise — word of mouth was how most folks knew of me. At one point I did have a website, but didn't utilize it much. I stayed very busy, especially in the summers, and always had a full schedule. I typically met with each student's teacher in order to get an idea of what the focus of my instruction/remediation should be. Becoming familiar with the student's learning style(s) was also very helpful. Often parents would share records and test results with me. After each tutoring session, I documented the student’s progress and gave parents written updates, as well as phone calls or emails as needed.
While my students and I certainly utilized technology and the internet often during our sessions, I never tutored online or for any tutoring company, so am not familiar with the curriculums or methods used in those settings.
Tutoring one on one was very enjoyable and rewarding. My students and I had fun, and grew quite fond of one another. The extra income was a bonus. I had to retire from tutoring due to a physically handicapping disease, and miss my students very much.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Consider the standard galvanic cell based on the following half reactions Cu2+ + 2e- Cu Ag+ + e- Ag The electrodes in this cell are Ag(s) and Cu(s). Does the cell potential increase, decrease, or...
-
Consider the galvanic cell based on the following halfreactions: b. Calculate ÎGo and K for the cell reaction at 25oC. c. Calculate cell at 25oC when [Zn2+] = 0.10 M and [Fe2+] = 1.0 Ã...
-
Consider the galvanic cell based on the following halfreactions: b. Calculate ÎGo and K for the cell reaction at 25oC. c. Calculate cell at 25oC when [Au3+] = 1.0 Ã 102 M and [Tl+] = 1.0...
-
Route Canal Shipping Company has the following schedule for aging of accounts receivable: AGE OF RECEIVABLES APRIL 30, 2001 a. Fill in column (4) for each month. b. If the firm had $1,440,000 in...
-
Link from the text Web site to the online Economic Report of the President and get annual data on real GDP and real investment. Calculate the growth rates of the two variables for each year since...
-
The following table lists the probability distribution for cash prizes in a lottery conducted at Lawsons Department Store: Prize ($) ................Probability $0...
-
How can sales promotion techniques be used to help the sales effort?
-
Special order pricing Shorewood Shoes Company makes and sells a variety of leather shoes for children. For its current mix of different models and sizes, the average selling price and costs per pair...
-
At December 31, 2022, the following information was available for E. Concord Company: ending inventory $32,000, beginning inventory $63,000, cost of goods sold $327,750, and sales revenue $370,000....
-
Deepa Dalal opened a free-standing radiology clinic. She had anticipated that the costs for the radiological tests would be primarily fixed, but she found that costs increased with the number of...
-
When the environment is contaminated by a toxic or potentially toxic substance (for example, from a chemical spill or the use of insecticides), the substance tends to disperse. How is this consistent...
-
In the electrolysis of a sodium chloride solution, what volume of H 2 (g) is produced in the same time it takes to produce 257 L Cl 2 (g), with both volumes measured at 50. C and 2.50 atm?
-
Two aspects that affect the form of opinion in an auditors report are a. The need to issue a modified report or an adverse report. b. The scope of the auditors work and the fairness of presentation...
-
When tasked with ventilation a fire fighter has numerous considerations to take into account before performing the task. List and defend 5 considerations that you take into account if and when...
-
How do developmental perspectives on emotional intelligence, grounded in attachment theory, social learning theory, and socioemotional development frameworks, illuminate the early childhood roots of...
-
Illustrate using examples of project management scenarios, the importance of the constraints of risk, quality, scope, time, cost, and resources on the final project schedule. Make reference to Tools...
-
After carefully reading The Great Inventory Correction case beginning on page 187 of the Simchi-Levi, et. al (2022) text and performing a thorough review of relevant literature, you will be assuming...
-
Consider the function U (p,b) = 2ln (p) + In (b) (a) Find the unconstrained maximum of the function (b) Now, suppose that the maximization is subject to the constraint 2p+36=24. What is the...
-
During basketball practice Shane made a jump shot, releasing a 0.60-kg basketball from his hands at a height of 2.0 m above the floor with a speed of 7.6 m/s. The ball swooshes through the net at a...
-
In your audit of Garza Company, you find that a physical inventory on December 31, 2012, showed merchandise with a cost of $441,000 was on hand at that date. You also discover the following items...
-
Compound F, a hydrocarbon with M + = 96 in its mass spectrum, undergoes reaction with HBr to yield compound G. Propose structures for F and G, whose 13 C NMR spectral data follow. Compound 1:...
-
3-Methyl-2-butanol has five signals in its 13C NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75 ?. Why are the two methyl groups attached to C3 nonequivalent? Making a molecular model should be...
-
A 13C NMR spectrum of commercially available 2, 4-pentanediol, shows five peaks at 23.3, 23.9, 46.5, 64.8, and 68.1 ?. Explain. CHCH-CHCH3 2,4-Pentanediol
-
Design a Turing Machine (TM) for the following language, give formal description of the TM and draw state diagram: {w|w is a string over the alphabet {0, 1} and number of 1's in w is divisible by 3}....
-
Jimmy John Hunter owns a small farm outside Winona and was getting very frustrated with turkey vultures that pester his sleeping livestock to the point where his chickens are laying fewer eggs. A few...
-
If the beginning cash balance is $ 5,000, the net cash inflow from operating activities is $20.000, net cash outflow from investing activities is (15,000) and net inflows from financing activities is...

Study smarter with the SolutionInn App


