Examine Figure 12-10 and state the phases that will be present in each stage if this process
Question:
Examine Figure 12-10 and state the phases that will be present in each stage if this process path is followed: (500°C, 20 wt% Mg) to (500°C, 80 wt% Mg) to (200°C, 80 wt% Mg).
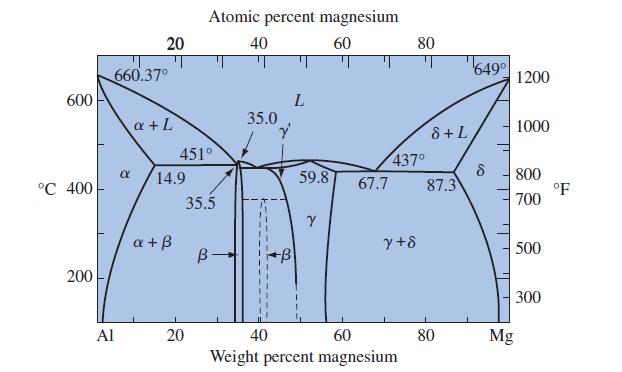
Transcribed Image Text:
600 °C 400 200 Al 20 660.37° α a + L 14.9 a + ß 451° Atomic percent magnesium 40 60 20 35.5 B- 35.0 Y 322 L 59.8 Y 67.7 437⁰ y+8 80 40 60 Weight percent magnesium 8 + L 87.3 80 649⁰ 8 Mg 1200 1000 800 700 500 300 °F
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
The phases include i High temperature melt which wil...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
What polarization mechanism will be present in (a) alumina; (b) copper; (c) silicon; and (d) barium titanate?
-
Figure P19.62 shows a thermodynamic process followed by 120 mg of helium. a. Determine the pressure (in atm), temperature (in C), and volume (in cm 3 ) of the gas at points 1, 2, and 3. Put your...
-
Examine Figure 8-12 to see that competitive firm C is not producing at all. Explain the reason why the profit maximizing output level for firm C is at q, = 0. What would happen to total industry cost...
-
Clara Hughes, who is pushing 5 0 , has medaled both in speedskating and road cycling ( and showing no signs of slowing down ) completed a training event where she biked 5 0 km east, stopped and rode...
-
Explain how the capital expenditures budget is different from the expense budget.
-
Elmo's Music carries a large inventory of guitars and other musical instruments. The store uses the FIFO method and a perpetual inventory system. Company records indicate the following for a...
-
What does the phrase theory of the case mean? Why is it important to develop a theory of the case in advance of trial?
-
Eclipse Construction Company is considering selling excess machinery with a book value of $280,000 (original cost of $400,000 less accumulated depreciation of $120,000) for $221,000, less a 5%...
-
Wearable technologies are here, and the applications for both personal and business use are truly inspiring. You can expect them to have a major impact on your everyday lives over the next decade....
-
The following trial balance of Sergei Asbrecht Co. does not balance. Each of the listed accounts has a normal balance per the general ledger. An examination of the ledger and journal reveals the...
-
Draw the eutectoid portion of the Fe- Fe 3 C phase diagram. Be sure to indicate all of the compositions and temperatures and write the relevant reaction.
-
Little Books Inc. recently reported $3 million of net income. Its EBIT was $6 million, and its tax rate was 40%. What was its interest expense? [Hint: Write out the headings for an income statement...
-
What ratio measures the firms degree of indebtedness? What ratios assess the firms ability to service debts?
-
Journalise the following transactions of a hypothetical firm during the period January 1 to January 31. January 11 January 2 January 3 January 4 January 6 January 6 January 6 January 10 January 12...
-
Para-xylene (p-xylene) is an important chemical intermediate in the manufacture of polyester fi bers. It is produced in high volume (around 109 kg/year). Consider a mixed stream of 60% m-xylene, 14%...
-
If the tax return or insurance claim is misstated, did Greg Larsen commit some form of financial statement fraud?
-
What accuracy certifications on the tax return and bank financial statement address providing materially inaccurate information?
-
Compare your recalculated ending inventory to that given to Sharptop Bank and to Southern Appalachian Insurance. Is either estimated ending inventory materially different from that reported on the...
-
A skeleton of Pine Florals' income statement appears as follows (amounts in thousands): Use the following ration data to complete Pine Florials' income statement: a. Inventory tunover was 4...
-
Data on weekday exercise time for 20 females, consistent with summary quantities given in the paper An Ecological Momentary Assessment of the Physical Activity and Sedentary Behaviour Patterns of...
-
Using estimates of rainfall, evaporation, and water consumption, the town engineer developed the following model of the water volume in the reservoir as a function of time where V is the water volume...
-
The volume V and paper surface area A of a conical paper cup are given by where r is the radius of the base of the cone and h is the height of the cone. a. By eliminating h, obtain the expression for...
-
A torus is shaped like a doughnut. If its inner radius is a and its outer radius is b, its volume and surface area are given by a. Create a user-defined function that computes V and A from the...
-
Several years ago, Westmont Corporation developed a comprehensive budgeting system for planning and control purposes. While departmental supervisors have been happy with the system, the factory...
-
Siblings, Aishwarya, Dhanya and Ganesh own properties around Malaysia and derive rental income from these properties. Listed below are the properties owned by them with the market value as at...
-
1. At an operational level in your own job role, how does each of the following impact you? Contents of national codes and standards that underpin regulatory requirements Reasons for food safety...

Study smarter with the SolutionInn App


