Explain why the development of a vapor pressure above a liquid in a closed container represents an
Question:
Explain why the development of a vapor pressure above a liquid in a closed container represents an equilibrium. What are the opposing processes? How do we recognize when the system has reached a state of equilibrium?
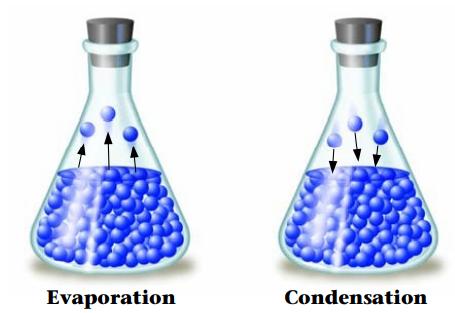
Transcribed Image Text:
Evaporation Condensation
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
Lets explain why the development of a vapor pressure above a liquid and a close container repr...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
How do we recognize when we are being manipulated by advertisers and avoid it?
-
Explain how groups develop. Trace the development of a group in which you have participated.
-
Why is the development of a marketing budget so important to the success of the marketing plan?
-
AASB 102 prohibits the use of the LIFO method. What is the argument against the use of LIFO?
-
Firms may seek the help of full-service or limited-service advertising agencies for their advertising. Describe each. What are the different departments of a full-service agency?
-
What is the primary distinction between financial accounting and managerial accounting?
-
Lucenay Interiors, a furniture store, was formed on January l, 2008, when lucenay issued common stock for \($400,000.\) Early in January, Lucena) made the following cash payments: a. \($100,000\) for...
-
Refer to the previous question. Suppose that each of the audit activities can be crashed by the amounts indicated in the following table. a. What is the earliest the audit could be completed and what...
-
Freeman Company purchased a tract of land on which is located a warehouse and a building. The cash purchase price was $252,000 plus $18,000 in fees connected with the purchase. The following...
-
Jefferson Animal Rescue is a private not-for-profit clinic and shelter for abandoned domesticated animals, chiefly dogs and cats. At the end of 2011, the organization had the following account...
-
Distinguish between the terms equilibrium constant and reaction quotient. When Q = K, what does this say about a reaction? When Q < K, what does this say about a reaction? When Q > K, what does this...
-
What is the difference between K and K p ? When does K = K p for a reaction? When does K K p for a reaction? If the coefficients in a reaction equation are tripled, how is the new value of K related...
-
Agilent Technologies, Inc., a diversified technology company, sells extended warranties for the products and services provided to customers, deferring the revenue until future recognition. The...
-
Engine oil at a rate of \(0.05 \mathrm{~kg} / \mathrm{s}\) flows through a \(3 \mathrm{~mm}\) diameter tube that is \(30 \mathrm{~m}\) long. The oil has an inlet temperature of \(40^{\circ}...
-
A capacitor initially has a charge of magnitude \(q\) on each plate. When a dielectric is inserted between the plates, the bound surface charge on the two dielectric surfaces facing the plates has a...
-
Explain why the value of pollution permits in one area of the country rose 20 percent per year, while in another remained unchanged from year to year? What would you expect to occur as a result of...
-
A capacitor connected to a battery initially holds a charge of \(+q\) on its positive plate and \(-q\) on its negative plate. The electric field between the plates is initially \(\vec{E}\). A...
-
Suppose two capacitor plates have an area of \(1 \mathrm{~cm}^{2}\) and are initially separated by \(2 \mathrm{~mm}\). Each plate holds \(5 \mu \mathrm{C}\) of charge. How much energy is required to...
-
Recently, a food retailer called Whole Foods sought to purchase Wild Oats, a competitor in the market for organic foods. When the Federal Trade Commission (FTC) sought to block this merger on...
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
Predict the 1H.NMR spectra of these compounds include the approximate chemical shift, multiplicity, and integral for each type ofhydrogen. CI b) CH;CHCH; ) C,CH,H c) CH,CH,OCH,CH3 CH2CH2NO2 f)...
-
How many different absorption bands would appear in the 13C-NMR spectra of thesecompounds? b) CH;CH,CH,CH,CH3 c) CH CCH,CH;CH,CH, d)
-
Assign the absorptions in the 13C-NMR spectra of these compounds to the appropriate carbons: (a) 1-Butanol; absorptions at 61.4, 35.0, 19.1, and 13.6 (b) Cyclohexanone; absorptions at 209.7, 41.9,...
-
The Step Company has the following Information for the year just ended: Sales in units Budget 15,000 Actual 14,000 Sales $150,000 $147,000 Less: Variable Expenses 90,000 82,600 Contribution Margin $...
-
Question 2 (2 marks) The year level coordinators for Years 7 to 11 at the school are Amy (A), Brian (B), Claire (C), Daisy (D) and Ellie (E). A faulty telephone system means that some of these...
-
On a certain farm alfalfa yields have been 4.3, 7.2, 5.6, and 3.6 tons per acre in the last 4 years. What is the expected value for the farm's alfalfa yield, if each past result is given equal weight...

Study smarter with the SolutionInn App


