Figure 9-29 shows a cylindrical riser attached to a casting. Compare the solidification times for each casting
Question:
Figure 9-29 shows a cylindrical riser attached to a casting. Compare the solidification times for each casting section and the riser and determine whether the riser will be effective.
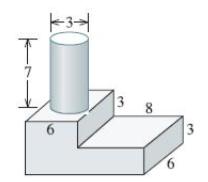
Transcribed Image Text:
K31 6 3 8 6 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The comparison is that If the riser is effective all sections sh...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Figure 9-23 shows a cylindrical riser attached to a casting. Compare the solidification times for each casting section and the riser and determine whether the riser will be effective?
-
Figure 9-30 shows a cylindrical riser attached to a casting. Compare the solidification times for each casting section and the riser and determine whether the riser will be effective. 8 k41 4 2 4
-
Can Brooklyn Co. determine whether currency options will be more or less expensive than a forward hedge when considering both hedging techniques to cover net payables in euros? Why or why not?
-
+9.33 x 10 C +91 Find the net force on 92. 0.180 m +4.22 x 10 C +92 0.230 m- -8.42 x 10 C 93 F = force exerted on q2 by 91 F3 = force exerted on 92 by 93 F = 10.9 N F3 = 6.04 N Remember: Forces...
-
Draw for Nation 2 a figure analogous to the top panel of Figure 7.5 under the following assumptions: (a) Nation 2 is now large enough to affect the relative commodity prices at which it trades. (b)...
-
For the vectors in Exercise 40, find ortha b and illustrate by drawing the vectors a, b, proj a b, and orth a b.
-
Justify what general factors you consider should be taken into account when recruiting salespeople for the positions described in the exercise. In particular, suggest how the performance of such...
-
Tried and Trues accountant (from Problem 1-1) suggests that monthly rent and hourly wage rate also be factored into the productivity calculations. Annandale pays the highest average wage at $6.75 an...
-
John Crossborder buys 1 share of Telmex at 140 pesos when thevalue of the peso is stated in dollars at $0.35. One yearlater, Telmex is selling for 160 pesos. If after 1 year thevalue of the pesos is...
-
On September 2, 2010, the Securities & Exchange Commission brought an action against Sujata Sachdeva, vice president of finance, and Koss senior accountant and subordinate, Julie Mulvaney, who...
-
A hollow cylindrical mold for casting aluminum ingots has a 300 mm inside diameter and is 2 m high. If the mold is filled with liquid aluminum at 935 K, what is the largest spherical cavity that may...
-
A 4-in. cube of a liquid metal is allowed to solidify. A spherical shrinkage cavity with a diameter of 1.49 in. is observed in the solid casting. Determine the percent volume change that must have...
-
Use the web to look up the percentage of dopant for a commercially available n-type semiconductor. Imagine that you were setting up a process for doping 1 metric ton of silicon with this dopant. (a)...
-
Over the winter months, Cullumber Co. pre-sells fertilizing and weed control lawn services to be performed from May through September, inclusive. If payment is made in full by April 1, a 5% discount...
-
On December 1, Jasmin Ernst organized Ernst Consulting. On December 3, the owner contributed $83,540 in assets to launch the business. On December 31, the company's records show the following items...
-
Dozier Company produced and sold 1,000 units during its first month of operations. It reported the following costs and expenses for the month: Direct materials Direct labor $ 89,000 $ 45,000 Variable...
-
Omira Cycle Parts manufactures components for motorcycle and off-road vehicles. The company's administrative and manufacturing operations share the company's only building. Eighty percent of the...
-
A company had the following assets and liabilities at the beginning and end of this year. Beginning of the year End of the year Assets $ 142,500 190,000 Liabilities $ 61,090 76,950 a. Owner made no...
-
Give the product of each of the following reactions: a. b. c. d. e. f. g. h. CH CH3 CH CH H2S04 CH,CH CHCH CH ,504 CH CH-CCH CH3 OH ' CH3O CH3OH 1. TsCl/pyridine 2. NaCEN CH3 OH (CH2CH2CH2hCuli CI
-
C- Consider the following scenario:- A supermarket needs to develop the following software to encourage regular customers. For this, the customer needs to supply his/her residence address, telephone...
-
Consider a molecule that can fluoresce from the S 1 state and phosphoresce from the T 1 state. Which is emitted at longer wavelength, fluorescence or phosphorescence? Make a sketch showing...
-
This problem can be worked with Equations 18-6 on a calculator or with the spreadsheet in Figure 18-5. Transferrin is the iron-transport protein found in blood. It has a molecular mass of 81 000 and...
-
The spreadsheet lists molar absorptivities of three dyes and the absorbance of a mixture of the dyes at visible wavelengths. Use the least-squares procedure in Figure 18-3 to find the concentration...
-
12. An amusement park ride swings riders around a 25 meter radius circle at a speed of 16 m/s. What is the centripetal acceleration in meters per second-squared felt by each rider? (Answer with 3...
-
For the image matrix given , use the Sobel horizontal edge detector to 1 2 -2 detect horizontal edges. Sobel matrix for horizontal edges is 0 0 104
-
What role does a resource allocation graph play in identifying potential deadlocks, and how can it be utilized to devise strategies for deadlock prevention ?

Study smarter with the SolutionInn App


