Given the thermodynamic data below, calculate S and S surr for the following reaction at 25
Question:
Given the thermodynamic data below, calculate ΔSο and ΔSsurr for the following reaction at 25οC and 1 atm:
![]()
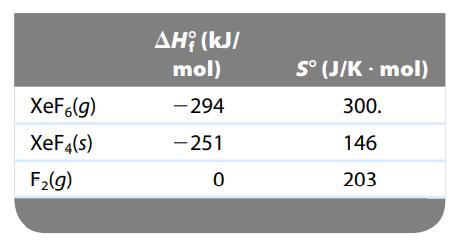
Transcribed Image Text:
XeF (g) XeF4(s) + F₂(8)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
To calculate S and Ssurr we need to use the equation ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
When 20g of CACO3 were put into 10 litre flask and heated to 800 C, 35% CACO, remained unreacted at equilibrium, Kp for decomposition of CACO3 is (A) 1.145 atm (B) 0.145 atm (C) 2.145 atm (D) 3.145...
-
Calculate the (E for the following reaction at 298 K 2H2(g) Og(g)- 2H2O(1)
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Fe3 + (aq) + Cu(s) Fe2+(aq) + Cu2+(aq) The equation is not balanced.
-
True or False: 1. Economists are not completely in agreement on what constitutes money for all purposes. 2. People use nontransaction accounts primarily because they generally pay higher interest...
-
The data in Figure 12.14 suggest an unemployment coefficient of approximately -1.0 in the Phillips curve. That is, the Phillips curve is n- n(-1) = -(1.0)(U - U*) . Assume the natural rate, U*, is 5...
-
The one-way commuting times from home to work for all employees working at a large company have a mean of 34 minutes and a standard deviation of 8 minutes. a. Using Chebyshevs theorem, find the...
-
What is the internal rate of return of the following cash flow diagram? a. 20 percent b. 18.2 percent c. 17.5 percent d. 15 percent $30 $31 0 1 2 3 $30 $15
-
The Central Valley Company has prepared department overhead budgets for budgeted-volume levels before allocations as follows: Management has decided that the most appropriate inventory costs are...
-
List the four main data definition language keywords and explain them with examples?
-
SOLO Pump Australia manufactures various pumps for niche industrial applications. The company has four main pumps (imaginatively named P1, P2, P3, and P4), which are assemblies of different...
-
For rubidium H vap = 69.0 kJ/mol at 686 C, its boiling point. Calculate S , q, w, and E for the vaporization of 1.00 mole of rubidium at 686 C and 1.00 atm pressure.
-
A reaction has K = 1.9 10 14 at 25C and K = 9.1 10 3 at 227C. Predict the signs for G o , H o , and S o for this reaction at 25C. Assume H o and S o do not depend on temperature.
-
In Fig. 4-41, a ball is launched with a velocity of magnitude 10.0m/s, at an angle of 50.0 o to the horizontal. The launch point is at the base of a ramp of horizontal length d 1 = 6.00 m and height...
-
An ice freezer behind a restaurant has a freon leak, releasing 43.21 g of C, H,F,Cl into the air every week. If the leak is not fixed, how many kilograms of fluorine will be released into the air...
-
Two small towns are adjacent to one another. Town A has a population of 500 people, while Town B has a population of 1200. Town A has a population growth rate of 4% per year. Town B's annual growth...
-
How could the issue have been mitigated based on the relationship between the organization and vendor? What strategies related to understanding the vendor (commitment, etc.) have helped? What...
-
Ella, a portfolio manager, considered several ways to invest USD15 million for one year. The data are as follows: USD interest: 7% per annum (p.a.) GBP interest: 10% p.a. Spot exchange rate:...
-
Answer the following two questions below. The Film link is located below. WATCH LINK https://digitalcampus-swankmp-net.libproxy.sdsu.edu/sdsu301489/play/98a88b1a7eefad09?referrer=lms when...
-
A steam power plant operates on the following cycle producing a net power of 25 MW. Steam enters the turbine at 16 MPa, 550oC and enters the condenser as saturated mixture at 10 kPa. Sub cooled...
-
An Atomic Energy Commission nuclear facility was established in Hanford, Washington, in 1943. Over the years, a significant amount of strontium 90 and cesium 137 leaked into the Columbia River. In a...
-
Would you expect a conjugated diyne such as 1, 3-butadlyne to undergo DielsAlder reaction with a dienophile explain.
-
Reaction of isoprene (2-methyl-1, 3-hutadiene) with ethyl propenoate gives a mixture of two Diels?Alder adducts. Show the structure of each, and explain why a mixture is formed. CO2CH2CH3
-
Rank the following dienophiles in order of their expected reactivity in the Diels?Alder reaction. CH CH NC NC CN c=C CH c=c c=C c=C NC CN
-
What are the trade-offs between query consistency and query isolation levels in distributed database systems, and how do different isolation levels, such as read committed, snapshot isolation, and...
-
Explain the Conflict theory of criminology
-
How do advanced query optimization techniques, such as cost-based optimization, query rewriting, and query hints, enhance the efficiency and performance of complex SQL queries in relational database...

Study smarter with the SolutionInn App


