Name the compounds in parts ad and write the formulas for the compounds in parts eh. a.
Question:
Name the compounds in parts a–d and write the formulas for the compounds in parts e–h.
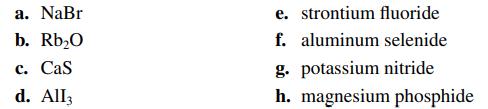
Transcribed Image Text:
a. NaBr b. Rb₂0 c. Cas d. All3 e. strontium fluoride f. aluminum selenide g. potassium nitride h. magnesium phosphide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
a NaBr Sodium bromide b RbO Rubidium oxi...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Write formulas for and name the binary hydrogen compounds of the second-period elements (Li to F). Describe how the physical and chemical properties of these compounds change from left to right...
-
Write expanded formulas for the following compounds and name them using the IUPAC system: a. (CH3)3CCH2CH2CH3 b. CH3(CH2)2CH3 c. (CH3)2CHCH2CH2CH3 d. CH3CCl2CF3 e. (CH2)4 f. CH3CH2CHFCH3 g. EtBr h....
-
Write the molecular and structural formulas for the compounds represented by the following molecular models: (a) (b) (c) (d) P F F F
-
1. If net profit is $47,025; depreciation is $2,200; accounts receivable increases $5,056; accounts payable increases $4,673; and inventory declines $850; what is the operating cash flow for the...
-
Pamello, Inc., an engineering consulting firm, uses the cash method of accounting and is a calendar year taxpayer. Compute the amount of Pamellos current year deductions for the following...
-
Supply file you used in this chapter. Modify the Comparative Summary Balance Sheet you created in the chapter (remember, you memorized it, so it is easy to recall) to include September, October, and...
-
Arlene Lavoie practiced law with a partnership for 10 years. Recently she opened her own law office, which she operates as a professional corporation. The name of the new entity is Arlene Lavoie,...
-
Wafers, Inc., processes silicon crystals into purified wafers and chips. Silicon crystals cost $60,000 per tank-car load. The process involves heating the crystals for 12 hours, producing 45,000...
-
3) Jamie makes a triangular pen to keep the puppies he just adopted. The lengths of the sides of the pen are 38 inches, 31 inches, and 35 inches. Determine the area of the pen. Round to the nearest...
-
A factory produces office chairs. According to the past data, the weekly demand has the following probability distribution. The selling price per chair is $120. In addition, the historical data...
-
Complete the following table. Atom/lon 50 Sn 25Mg+ 2+ 56 Fe+ 120S 79 32 Se 34- 35C1 53 Cu Protons Neutrons Electrons
-
For carbon-14 and carbon-12, how many protons and neutrons are in each nucleus? Assuming neutral atoms, how many electrons are present in an atom of carbon-14 and in an atom of carbon-12?
-
Under the fair value method, when and how does the investor company measure revenue?
-
Develop and solve a future-value, a present-value, a future value of an annuity, and a present value of an annuity problem. Establish the three known variables in each problem and solve for the...
-
Quantitative Risk Assessment. Assume the following information for an IT project: Calculate the overall risk factor for this project. Would you assess this level of risk as low, moderate, or high?...
-
Assume that your senior managers have allocated a labor cost of $30,000 for your project. Using the following data, calculate the fully loaded labor costs for each person. What is the total cost? Can...
-
You have a partial network for your project, and you are about to conduct a forward pass through it. Explain why the early start (ES) for activity G is 23 days, not 19 days. Remember, activity G is a...
-
If households and firms change from expecting mild inflation to expecting mild deflation, how will the Phillips curve shift? Draw an output gap Phillips curve graph to illustrate your answer.
-
Brady Furniture Corp. is nationally recognized for making high-quality products. Management is concerned that it is not fully exploiting its brand power. Brady's production managers are also...
-
The following data are supplied for the common stocks of Nikola Corporation, Tesla, Inc. and General Motors: Nikola Corp (NKLA) Tesla Inc. (TSLA) Close Price ($) Close Price ($) 67.53 30.00 40.81...
-
The 1980s saw reports of fH (SiH2) ranging from 243 to 289 k] mol-1. For example, the lower value was cited in the review article by R. Walsh (Ace. Chem. Res. 14,246 (1981)); Walsh later leant...
-
Given that tG = -212.7 kJ mol-1 for the reaction in the Daniell cell at 25C, and b (CuS04) = 1.0 x 10-3 mol kg-i and b (ZnS04) = 3.0 x 10-3 mol kg-I, calculate (a) The ionic strengths of the...
-
Although the hydrogen electrode may be conceptually the simplest electrode and is the basis for our reference state of electrical potential in electrochemical systems, it is cumbersome to use....
-
1. Identifield and explain 3 causes of stress and 3 techniques to overcome stress ? 2. examine worksite stress management programmers more likely to be effective.
-
Bright Books is a bookstore chain established 10 years ago in northern Michigan. Bright Books has had great success in Michigan and Ohio and is beginning to expand into other states. Most recently,...
-
Design a synchronous counter using D flip-flops such that the count sequence of the counter follows the digits in . Each state must be 4-bit. Follow the steps below: (i) (ii) (iii) (iv) 137836...

Study smarter with the SolutionInn App


