Suppose K = 4.5 10 -3 at a certain temperature for the reaction If it is
Question:
Suppose K = 4.5 × 10-3 at a certain temperature for the reaction
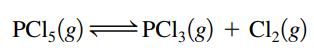
If it is found that the concentration of PCl5 is twice the concentration of PCl3, what must be the concentration of Cl2 under these conditions?
Transcribed Image Text:
PC15 (8) PC13 (8) PC13(g) + Cl₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
Solution The concentratio...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The following equilibrium pressures were observed at a certain temperature for the reaction N2(g) + 3H2(g) 2NH3(g) PNH3 = 3.1 10-2 atm PN2 = 8.5 10-1 atm PH2 = 3.1 10-3 atm Calculate the value...
-
It is found that a 6.00-m segment of a long string contains four complete waves and has a mass of 180 g. The string is vibrating sinusoidally with a frequency of 50.0 Hz and a peak-to-valley distance...
-
Suppose that following the CME it is found that at an altitude of 2500 m the electric field has magnitude 1300 N/C and at an altitude of 1500 m the magnitude is 3600 N/C. In both cases the direction...
-
Consider the pooled t variable Tp from part (b) of the previous exercise. a. Use this t variable to obtain a pooled t confidence interval formula for 1 2 . b. The article Effect of Welding on a...
-
What is a supply chain, and how is it different from a channel of distribution?
-
AnderSet Laboratories produces rough lenses that will ultimately be ground into precision lenses for use in laboratory equipment. The company has developed the following thickness measures, based on...
-
Which of the following is improper when using probability-proportional-to-size sampling? a. Combining negative and positive dollar error items. b. Using a sample selection technique in which the same...
-
Incomplete inventory and income statement data for Gator Corporation follow. Determine the missingamounts. Cost of Goods Sold Cost of Goods Manufactured $20,000 Beginning Finished Ending Finished...
-
On January 1, 2020, Stream Company acquired 20 percent of the outstanding voting shares of Q-Video, Inc., for $702,000. Q-Video manufactures specialty cables for computer monitors. On that date,...
-
A statement of cash flows for Friendly Markets, Inc., for 20X1 appears below. Operations (1) Net income $ 1,161,442 Adjustments for noncash transactions: (2) Depreciation and amortization 496,106 (3)...
-
At a certain temperature, K = 9.1 10 -4 for the reaction Calculate the concentrations of Fe 3+ , SCN - , and FeSCN 2+ in a solution that is initially 2.0 M FeSCN 2+ . 3+ FeSCN2+ (aq) Fe+ (aq) + SCN-...
-
Old-fashioned smelling salts consist of ammonium carbonate, (NH 4 ) 2 CO 3 . The reaction for the decomposition of ammonium carbonate is endothermic. Would the smell of ammonia increase or decrease...
-
Research and write a short paper (two pages maximum) on the skills needed to be a successful project manager.
-
Elle was 65 as of January 1st 2022. Her RRIF was established in 2020. On January 1, 2022, Elle's RRIF contained assets valued at $210,000, however, due to market fluctuations - her portfolio was...
-
What qualities and characteristics do you think a good leader must possess?
-
Analyze how the current healthcare financing system is affecting access to basic healthcare services, quality of care and well-being of the nation?
-
What is international diversification? Discuss international investment strategies based on the textbook? What are the hedging strategies we can use to offset the currency risk involved in...
-
Does PE acquisition of physician practices result in higher or lower healthcare and spending utilization? Why do you think that is?
-
What technique could Becquerel and others have used to determine that a rays are positively charged, b rays negatively charged, and g rays uncharged? Explain how they could find that a rays have a...
-
Briefly describe the following types of group life insurance plans: a. Group term life insurance b. Group accidental death and dismemberment insurance (AD&D) c. Group universal life insurance d....
-
When 0.575 g of monosodium glutamate (MSG) is dissolved in 10.0 mL of water and placed in a sample cell 10.0 cm in length, the observed rotation at 20C (using the D line of sodium) is +1.47....
-
When 0.095 g of cholesterol is dissolved in 1.00 mL of ether and placed in a sample cell 10.0 cm in length, the observed rotation at 20C (using the D line of sodium) is -2.99. Calculate the specific...
-
When 1.30 g of menthol is dissolved in 5.00 mL of ether and placed in a sample cell 10.0 cm in length, the observed rotation at 20C (using the D line of sodium) is +0.57. Calculate the specific...
-
Is China's rapid economic growth, spurred by Deng Xiaoping's reforms in the 80s, sustainable in the long term, or does the current threat of deflation indicate potential risks that could undermine...
-
The market for Big Macs in Canada is initially in equilibrium with supply and demand curves of the usual shape. Whoppers are a close substitute for Big Macs; Fries and Big Macs are complements. Use...
-
The COVID-19 pandemic has adversely affected the lives of people around the world in millions of ways. Due to this severe epidemic, all countries in the world have been affected by all aspects,...

Study smarter with the SolutionInn App


