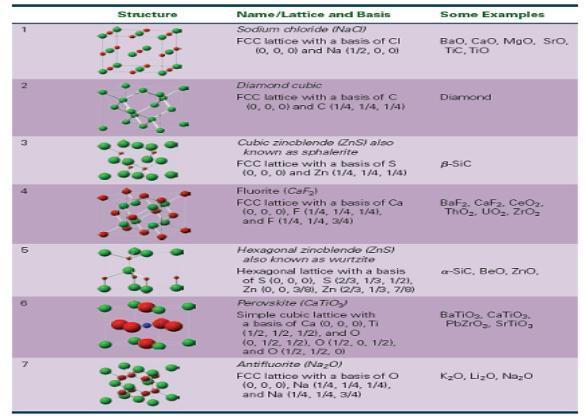
Table 15-4 describes the cubic zincblende structure as a face-centered cubic lattice with a basis of S
Question:
Table 15-4 describes the cubic zincblende structure as a face-centered cubic lattice with a basis of S (0, 0, 0) and Zn (1/4, 1/4, 1/4). Using S as one ion of the basis, give three other equivalent descriptions for the position of the Zn ion.
Transcribed Image Text:
Structure Name/Lattice and Basis Sodium chloride (NaC)) FCC lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 01 Diamond cubic FCC lattice with a basis of C (0, 0, 0) and C (1/4, 1/4, 1/4) Cubic zincblende (ZnS) also known as sphalerite FCC lattice with a basis of S (0, 0, 0) and Zn (1/4, 1/4, 1/4) Fluorite (CaF₂ FCC lattice with a basis of Ca (0, 0, 0), F (1/4, 1/4, 1/4), and F (1/4, 1/4, 3/4) Hexagonal zincblende (ZnS) also known as wurtzite Hexagonal lattice with a basis of S (0, 0, 0), S (2/3, 1/3, 1/2), Zn (0, 0, 3/0), Zn (2/3, 1/3, 7/8) Perovskite (CaTiO₂) Simple cubic lattice with a basis of Ca (0, 0, 0), Ti (1/2, 1/2, 1/2), and O (0. 1/2, 1/2). O (1/2, 0. 1/2). and O (1/2, 1/2, 0) Antifluorite (Na₂O) FCC lattice with a basis of O (0, 0, 0), Na (1/4, 1/4, 1/4). and Na (1/4, 1/4, 3/4) Some Examples Bao, Cao. MaO, SIO, TIC, TIO Diamond B-Sic BaF2. CaF₂. CeO₂. ThO₂. UO₂. ZrO₂ a-SiC, BeO, ZnO, BaTiO₂, CaTiO₂. PbZrO₂. SrTiO₂ K₂O, Li₂O, Na₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
Solution The three include i Assign each atom a Cartesian ...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 0). Using Na (0, 0, 0) as one ion of the basis, give three equivalent...
-
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 0). Using Cl (0, 0, 0) as one ion of the basis, give three equivalent...
-
Using Table 1.3, determine what charge the ion will carry when each of the following elements reacts to form an ionic compound: Al, Li, S, and O. Table 1.3 Table 1.3 Valence Electrons of the First 18...
-
Explain why b x = e x ln b .
-
A community health status survey obtained the following demographic information from the respondents: Compute the relative frequency and cumulative relative frequency of the age groups. Age 18 to 29...
-
The 20X5 records of Jax Company provided the following data related to its non-contributory, defined benefit pension plan (in \(\$\) thousands): Required: 1. Compute \(20 \times 5\) pension expense....
-
On April 30, 2010, Citibank loaned $100,000 to Grant Hughes on a one-year, 6% note. Requirements 1. Compute the interest for the years ended December 31,2010 and 2011, on the Hughes note. 2. Which...
-
A 1250-kg wrecking ball is lifted to a height of 12.7 m above its resting point. When the wrecking ball is released, it swings toward an abandoned building and makes an indentation of 43.7 cm in the...
-
What role does leadership play in shaping and sustaining organizational culture, and how do different leadership styles and behaviors impact cultural alignment, employee morale, and organizational...
-
While performing an audit of a public company, the auditors discovered material illegal acts and resigned due to the client's refusal to disclose them. The auditors' reason for resignation should be...
-
If a face-centered cubic crystal structure has all of its tetrahedral sites filled with atoms that fit perfectly into each tetrahedral site, what is its atomic packing factor?
-
Convert the properties of Al 2 O 3 from Table 15-3 into values with SI units. Table 15-3 Mechanical properties of selected advanced ceramics Flexural Strength (psi) Material AlO3 SiC (sintered) Si3N4...
-
Color conversion. Several different formats are used to represent color. For example, the primary format for LCD displays, digital cameras, and web pages, known as the RGB format, specifies the level...
-
Voltage fluctuation is given as the reason for \(80 \%\) of all defaults in nonstabilized equipment in a plant. Use the formula for the binomial distribution to find the probability that voltage...
-
If the probability is 0.40 that steam will condense in a thin-walled aluminum tube at 10 atm pressure, use the formula for the binomial distribution to find the probability that, under the stated...
-
A quality-control engineer wants to check whether (in accordance with specifications) \(95 \%\) of the electronic components shipped by his company are in good working condition. To this end, he...
-
Which conditions for the binomial distribution, if any, fail to hold in the following situations? (a) The number of persons having a cold at a family reunion attended by 30 persons. (b) Among 8...
-
An engineering student correctly answers \(85 \%\) of all questions she attempts. What is the probability that the first incorrect answer was the fourth one?
-
Consider the following data for two products of Gitano Manufacturing. Required 1. Using direct labor hours as the basis for assigning overhead costs, determine the total production cost per unit for...
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
An object starts with an initial velocity of 3 m/s at t = 0, and it accelerates with an acceleration of a(t) = 7t m/s 2 . Find the total distance the object travels in 4 s.
-
The equation for the voltage (t) across a capacitor as a function of time is where i(t) is the applied current and Q 0 is the initial charge. A certain capacitor initially holds no charge. Its...
-
A certain objects acceleration is given by a(t) = 7t sin 5t m/s 2 . Compute its velocity at t = 10 s if its initial velocity is zero.
-
pints 3 Maria Am Corporation uses the weighted-average method of process costing. The Baking Department is one of the processing departments in its strudel manufacturing facility. In June in the...
-
The State Public Works Division consists of the Administrator, the State Public Works Board, the Public Works Section, and the Buildings and Grounds Section. The State Public Works Board consists of...
-
Explain the leadership styles, attitudes, behaviors, and characteristics that you think may be causing disengaged, unmotivated, and unproductive employees. provide sources/authors.

Study smarter with the SolutionInn App


