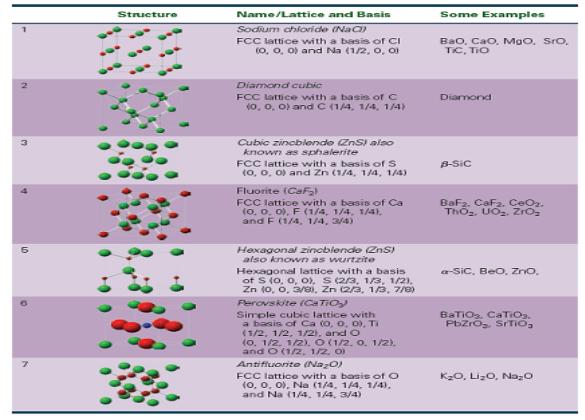
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl
Question:
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 0). Using Cl (0, 0, 0) as one ion of the basis, give three equivalent descriptions for the position of the Na ion.
Transcribed Image Text:
Structure Name/Lattice and Basis Sodium chloride (NaC)) FCC lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 01 Diamond cubic FCC lattice with a basis of C (0, 0, 0) and C (1/4, 1/4, 1/4) Cubic zincblende (ZnS) also known as sphalerite FCC lattice with a basis of S (0, 0, 0) and Zn (1/4, 1/4, 1/4) Fluorite (CaF₂ FCC lattice with a basis of Ca (0, 0, 0), F (1/4, 1/4, 1/4), and F (1/4, 1/4, 3/4) Hexagonal zincblende (ZnS) also known as wurtzite Hexagonal lattice with a basis of S (0, 0, 0), S (2/3, 1/3, 1/2), Zn (0, 0, 3/0), Zn (2/3, 1/3, 7/8) Perovskite (CaTiO₂) Simple cubic lattice with a basis of Ca (0, 0, 0), Ti (1/2, 1/2, 1/2), and O (0. 1/2, 1/2). O (1/2. 0. 1/2). and O (1/2, 1/2, 0) Antifluorite (Na₂O) FCC lattice with a basis of O (0, 0, 0), Na (1/4, 1/4, 1/4). and Na (1/4, 1/4, 3/4) Some Examples Bao, Cao. MaO, SIO, TIC, TIO Diamond B-Sic BaF2. CaF₂. CeO₂. ThO₂. UO₂. ZrO₂ a-SiC, BeO, ZnO, BaTiO₂, CaTiO₂. PbZrO₂. SrTiO₂ K₂O, Li₂O, Na₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
The position of the Na ion can be described in three diffe...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 0). Using Na (0, 0, 0) as one ion of the basis, give three equivalent...
-
Table 15-4 describes the cubic zincblende structure as a face-centered cubic lattice with a basis of S (0, 0, 0) and Zn (1/4, 1/4, 1/4). Using S as one ion of the basis, give three other equivalent...
-
Let Find x , y , xy , and yx , and state the domain for each partial derivative. f(x, y) = [y, y = 0 1-y, y < 0.
-
Use the COUNTIF function to construct a frequency distribution of the types of loans in the Excel file Credit Risk Data and develop a column chart to express the results visually.
-
What kind of mass movements might be expected in areas underlain by limestone or dolomite, as in Florida?
-
Water flows through the pipe shown in Fig. P8.87. Determine the net tension in the bolts if minor losses are neglected and the wheels on which the pipe rests are frictionless. Figure P8.87 80 mm 3.0...
-
Prepare a bank reconciliation for Bend Company at January 31, 2012, using the information shown. 1. Cash per the accounting records at January 31 amounted to $228,909; the bank statement on this same...
-
Tamarisco A partir del 1 de enero de 2025, Industries tena una patente registrada en sus libros. Esta patente tena un valor contable de 480.000 dlares y una vida til restante de 8 aos. Durante 2025,...
-
Preble Company manufactures one product. Its variable manufacturing overhead is applied to production based on direct labor-hours and its standard cost card per unit is as follows: Direct material: 4...
-
If a face-centered cubic crystal structure has all of its tetrahedral sites filled with atoms that fit perfectly into each tetrahedral site, what is its atomic packing factor?
-
Convert the properties of Al 2 O 3 from Table 15-3 into values with SI units. Table 15-3 Mechanical properties of selected advanced ceramics Flexural Strength (psi) Material AlO3 SiC (sintered) Si3N4...
-
(a) Define atomic number and mass number. (b) Which of these can vary without changing the identity of the element?
-
In factor markets, what is the primary resource that is bought and sold? Explain briefly
-
How study examined levels of gas prices and carbon monoxide levels for a sample of municipalities.? Explain briefly
-
Think critically about various perspectives on the current event or issue and how they relate to social work practice. What steps can social workers take to address oppression?
-
What is the velocity when the price level is 110, RGDP is 7200, and M is 660? Additional comments: the answer is 12, just need to know how to solve it. What is the velocity when the price level is...
-
How does blood quantum requirements and tribal enrollment impact where a Native person (and/or their children) might live? Explain briefly
-
Specify the group of the periodic table in which each of the following elements is found: (a) [Ne]3s1 , (b) [Ne]3s23p3 , (c) [Ne]3s23p6 , (d) [Ar]4s23d8.
-
After graduating from college and working a few years at a small technology firm. Preet scored a high-level job in the logistics department at Amex Corporation. Amex sells high-quality electronic...
-
A certain object has a mass of 100 kg and is acted on by a force f(t) = 500[2 e t sin(5t)] N. The mass is at rest at t = 0. Determine the objects velocity at t = 5 s.
-
The equation for the voltage (t) across a capacitor as a function of time is where i(t) is the applied current and Q 0 is the initial charge. Suppose that C = 10 7 F and that Q 0 = 0. Suppose the...
-
A rockets mass decreases as it burns fuel. The equation of motion for a rocket in vertical flight can be obtained from Newton s law, and it is where T is the rockets thrust and its mass as a function...
-
A company is faced with the decision of whether to lease or purchase new equipment. The equipment can be leased at an annual cost of $210,000 or purchased for $560,000. The equipment has an expected...
-
Braverman Company has two manufacturing departments-Finishing and Fabrication. The predetermined overhead rates in Finishing and Fabrication are $28.00 per direct labor-hour and 120% of direct...
-
A stock had returns of 28%, 1%, and 13% over the previous 3 years. The average return on this stock over the 3-year period was 14%. What was the variance of this stock's returns over the previous 3...

Study smarter with the SolutionInn App


