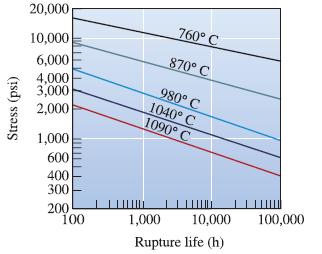
Using the data in Figure 7-26 for an ironchromium- nickel alloy, determine the activation energy Qr and
Question:
Using the data in Figure 7-26 for an ironchromium- nickel alloy, determine the activation energy Qr and the constant m for rupture in the temperature range 980 to 1090°C.
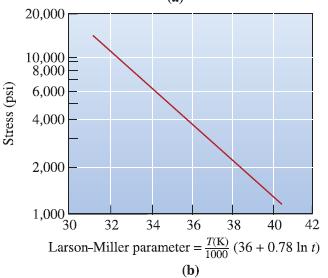
Transcribed Image Text:
Stress (psi) 20,000 10,000 6,000 4,000 3,000 2,000 1,000 600 400 300 200 100 760° C 870° C 980° C 1040° C 1090° C 1,000 10,000 Rupture life (h) 100,000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
The activation energy Qr HcT where Hc RT ...View the full answer

Answered By

Willis Omondi
Hi, I'm Willis Omondi, a proficient and professional academic writer. I have been providing high-quality content that best suits my clients and completing their work within the deadline. All my work has been 100% plagiarism-free, according to research from my services, especially in arts subjects and many others
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
7. Point P is located 1.7 m from fixed point charge q. The electric field E at point Pis 8.6 x 10-2 (1 point) N/C, directed away from the point charge. What is the magnitude of q? Let the...
-
Using the data in Solved Problem 10.1, prepare an activity chart like the one in the solved problem, but a second gas man also delivers 44 litres. In solved problem 10.1 Process Chart Summary O...
-
Using the data in BE21-4, compute equivalent units of production for materials and conversion costs, assuming materials are entered at the beginning of the process.
-
The management of a New York area investment firm wants to find out about the investment needs of its existing customers, for which it has an extensively detailed list, as a function of their...
-
Distinguish among a derivative action, dissent and oppression. Explain when it would be appropriate to use each of them.
-
Provide some examples of mental leaps that entrepreneurs have taken.
-
The homeowner decides to hire you to design a system for her. She has arranged with a local solar supplier for the following equipment. Specify the system and provide a line diagram. The owner of a...
-
Boreki Enterprise has the following 10 items in inventory. Theodore Boreki asks you, a recent OM graduate, to divide these items into ABC classification. (a) Develop an ABC classification system for...
-
< Break-Even Units, Contribution Margin Ratio, Multiple-Product Breakeven, Margin of Safety, Degree of Operating Leverage Jellico Inc.'s projected operating income (based on sales of 450,000 units)...
-
Brookhurst Company (a U.S.-based company) established a subsidiary in South Africa on January 1, Year 1, by investing 300,000 South African rand (ZAR) when the exchange rate was US$0.09/ZAR 1. On...
-
Approximate the temperature at which creep deformation becomes an important consideration for each of the following metals: tin, molybdenum, iron, gold, zinc and chromium.
-
What is the difference between failure of a material by creep and that by stress rupture?
-
What are the HRM implications of employing nonstandard workers in the gig economy?
-
Benjamin DeCosta, CFA has gathered selected year-end balance sheet information (all amounts in millions) for Palantir Technologies (PLTR): Total assets $11,050 Total liabilities $9,500 Preferred...
-
The accountant's work sheet: Question 25 options: serves as the basis for completing the adjusting process. lays the groundwork for formal financial statement preparation. assists in closing at the...
-
Kim and John separated 3 years after John's mother, Kathy passed away. When Kathy died, she left him $300,000 through her will. In which scenario would Kim be entitled to the inheritance? None of the...
-
Upon the conclusion of a 100,000-square-foot office building project, Casa Loma Development sold the property to Blue Jays Group. Subsequent to this transaction, Casa Loma Development and Blue Jays...
-
answered. Problem14This problem is current being displayed. Problem15 Question Content AreaStephen makes and sells cakes through bakeries. He knows he must sell 450 cakes a month to break even. Every...
-
a. Propose a mechanism for the following reaction: b. To what class of terpene does the starting material belong? Mark off the isoprene units in the starting material. H. d H CH CH3 CH3 CH CH3 CH3...
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
Specifications for lactated Ringers solution, which is used for intravenous (IV) injections, are as follows to reach 100. mL of solution: 285315 mg Na + 14.117.3 mg K + 4.96.0 mg Ca 2+ 368408 mg Cl -...
-
You have a solution of two volatile liquids, A and B (assume ideal behavior). Pure liquid A has a vapor pressure of 350.0 torr and pure liquid B has a vapor pressure of 100.0 torr at the temperature...
-
A solid mixture contains MgCl 2 and NaCl. When 0.5000 g of this solid is dissolved in enough water to form 1.000 L of solution, the osmotic pressure at 25.0 C is observed to be 0.3950 atm. What is...
-
1. Define human resource (HR) management and explain how it relates to the management process. Cite examples of the application of these concepts, preferably from personal, professional experience.
-
Mickley Corporation produces two products, Alpha6s and Zeta7s, which pass through two operations, Sintering and Finishing. Each of the products uses two raw materialsX442 and Y661. The company uses a...
-
Managers use CVP concepts to perform sensitivity analysis. Sensitivity analysis is a "what-if" technique that asks what will happen to a company's breakeven or target profit if sales price, costs, or...

Study smarter with the SolutionInn App


