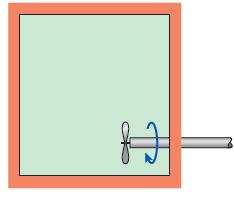
A substance is contained in a well-insulated rigid container that is equipped with a stirring device, as
Question:
A substance is contained in a well-insulated rigid container that is equipped with a stirring device, as shown in Fig. P4–30. Determine the change in the internal energy of this substance when 15 kJ of work is applied to the stirring device.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
A substance is contained in a wellinsulated rigid container tha...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
A 15 L tank contains 1 kg of R-12 refrigerant at 100oC. It is heated until the temperature of the refrigerant reaches 150oC. Determine the change in. (a) Internal energy (U). (b) Entropy, (S). Use...
-
A rigid container equipped with a stirring device contains 2.5 kg of motor oil. Determine the rate of specific energy increase when heat is transferred to the oil at a rate of 1 W, and 1.5 W of power...
-
In a refrigerator, 2.00 mol of an ideal monatomic gas are taken through the cycle shown in the figure. The temperature at point A is 800.0 K. (a) What are the temperature and pressure at point D? (b)...
-
Discuss why marketing management is important to an organization, and how corporate and business strategy analysis gives an organization a competitive advantage.
-
The owner of a restaurant serving Continental-style entrees has the business objective of learning more about the patterns of patron demand during the Friday-to-Sunday weekend time period. Data were...
-
A review of the ledger of Bellingham Accountants at December 31, 2020, produces the following data pertaining to the preparation of annual adjusting entries. 1. Prepaid Insurance 10,340. The company...
-
Find the statistical autocorrelation function of the random process \[ U(t)=a_{1} \cos \left(2 \pi v_{1} t-\Phi_{1} ight)+a_{2} \cos \left(2 \pi v_{2} t-\Phi_{2} ight) \] where \(a_{1}, a_{2},...
-
The vice president of operations if Avigi IQ Company is evaluating the performance of two division organized as investment centers. Invested assets and condensed income statement data for the past...
-
Mid-South Auto Leasing leases vehicles to consumers. The attraction to customers is that the company can offer competitive prices due to volume buying. On September 30, 2024, the company leased a...
-
There is a lottery with n coupons and n people take part in it. Each person picks exactly one coupon. Coupons are numbered consecutively from 1 to n, n being the maximum ticket number. The winner of...
-
Complete each line of the following table on the basis of the conservation of energy principle for a closed system. W. E, E, e2- e; kJ/kg m out kJ kJ kJ kJ kg 280 1020 550 860 3 5 -350 130 260 -150...
-
A rigid 1-ft 3 vessel contains R-134a originally at 20F and 27.7 percent quality. The refrigerant is then heated until its temperature is 100F. Calculate the heat transfer required to do this. Heat...
-
The expired cost with respect to a fixed asset is referred to as ______ expense.
-
Using any software tool of your choice (e.g., Visio, PowerPoint, MS Word, yED, Lucidchart, etc.) create, draw and submit... OPTION #1 A data flow diagram for an ATM system OR OPTION #2 A Level 0 DFD...
-
4. An object of mass 0.2 kg is hung from a spring whose spring constant is 80 N/m. The object is subject to a resistive force given by -by, where v is its velocity in meters per second. a. Set up the...
-
Q7. What is an isochoric process?
-
Lola is purchasing property worth $1,060,000 with a down payment of $212,000 and quarterly payments at the end of every three months for 30 years. If the interest rate of 2.07% compounded...
-
Duke Energy ( DUK ) is an electric utility company that provides electric power to 10 million customers in the U.S. specifically in the Southeast and Midwest. DUK will pay a dividend this coming year...
-
La Mode Design Inc., a high-fashion women's dress manufacturer, is planning to market a new cocktail dress for the coming season. La Mode Design Inc. supplies retailers in Toronto, Montreal, and the...
-
An interest bearing promissory note for 90 days at 5.6% p.a. has a face value of $120,000. If the note is discounted 20 days after the issue date at a rate of 6.8% p.a., calculate the amount of...
-
Saturated steam coming off the turbine of a steam power plant at 40oC condenses on the outside of a 3-cmouter- diameter, 35-m-long tube at a rate of 130 kg/h. Determine the rate of heat transfer from...
-
Water in a 5 - cm-deep pan is observed to boil at 98oC. At what temperature will the water in a 40 - cm-deep pan boil? Assume both pans are full of water.
-
A cooking pan whose inner diameter is 20 cm is filled with water and covered with a 4-kg lid. If the local atmospheric pressure is 101 kPa, determine the temperature at which the water starts boiling...
-
A consultant advises that a fund pays out $100,000 at the end of 5 years. Provided $12,000 is invested in the fund at the start of each year, beginning immediately and continuing up to and including...
-
Durco Automotive needs a $2 million balance in its contingency fund 3 years from now. The chief financial officer (CFO) wants to know how much to deposit now into Durco's high-yield investment...
-
3. Create a chart for gross margin less R&D and capital costs calculations. a. Calculate gross margin less R&D and capital costs for options A and B. b. Graph total gross margin less R&D and capital...

Study smarter with the SolutionInn App


