On a summer day in New Orleans, Louisiana, the pressure is 1 atm: the temperature is 32C;
Question:
On a summer day in New Orleans, Louisiana, the pressure is 1 atm: the temperature is 32°C; and the relative humidity is 95 percent. This air is to be conditioned to 24°C and 60 percent relative humidity. Determine the amount of cooling, in kJ, required and water removed, in kg, per 1000 m3 of dry air processed at the entrance to the system.
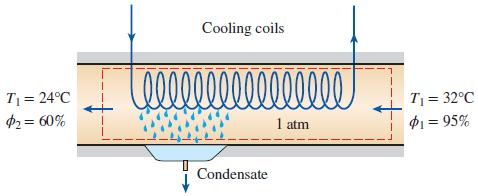
Cooling coils 00000000 T = 24°C $2= 60% T = 32°C P1 = 95% 1 atm Condensate
Step by Step Answer:
Air is cooled and dehumidified at constant pressure The amount of water removed from the air and the ...View the full answer



Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Related Video
In this video, the concept of pressure has been explained. The bottle is filled up with water, remove the pipe, and now you can see the clear flow of water from the holes due to liquid pressure.You can see that the second layer is holding the weight of the first layer and the third layer holding the weight of both upper layers, that’s why liquid flows out wider from the third as compared to the first two.At the first hole, water doesn’t roll along the walls; it flows out at an angle.At the second hole, you’ll see that the water flows out at a wider angle as compared to the first hole.At the third hole, we can see that the flow of water is much wider/greater than the other holes.This proves that the pressure at the bottom of the bottle is much more than the pressure at the top layers. P = ????gh
Students also viewed these Engineering questions
-
Reconsider Prob. 1482. How far will the temperature of the humid air have to be reduced to produce the desired dehumidification? Data From Reconsider Prob. 1482: On a summer day in New Orleans,...
-
During a summer day in El Paso, Texas, outdoor air is at 1 atm, 40C, and 20 percent relative humidity. Water at 20C is evaporated into this air to produce air at 25C and 80 percent relative humidity....
-
During a summer day in Phoenix, Arizona, the air is at 1 atm, 110oF, and 15 percent relative humidity. Water at 70oF is evaporated into this air to produce air at 75oF and 80 percent relative...
-
The following items were shown on the balance sheet of Herman Corporation on December 31, 2010: Stockholders Equity Paid-In Capital Capital Stock Common stock , $5 par value, 360,000 shares...
-
An observed frequency distribution is as follows: Number of successes 0 1 2 3 Frequency 89 133 52 26 a. Assuming a binomial distribution with n = 3 and p = 1/3, use the binomial probability...
-
On January 2, 2019, Phillips Company purchased 80% of Sanchez Company and 90% of Thomas Company for $225,000 and $168,000, respectively. Immediately before the acquisitions, the balance sheets of the...
-
An inward flow radial turbine (see Fig. P5.89) involves a nozzle angle, \(\alpha_{1}\), of \(60^{\circ}\) and an inlet rotor tip speed, \(U_{1}\), of \(6 \mathrm{~m} / \mathrm{s}\). The ratio of...
-
The comparative balance sheets for Strackman Lux Company as of December 31 are presented below. Additional information:1. Operating expenses include depreciation expense of $42,000 and charges from...
-
Which do you think are more important to emphasize in a literature review, the opinions of experts in the field or related studies and why? Describe the kind of behaviours that individual team...
-
The blood bank wants to determine the least expensive way to transport available blood donations from Pittsburg and Staunton to hospitals in Charleston, Roanoke, Richmond, Norfolk, and Suffolk. The...
-
Atmospheric air from the inside of an automobile enters the evaporator section of the air conditioner at 1 atm, 27C, and 50 percent relative humidity. The air returns to the automobile at 10C and 90...
-
Repeat Prob. 1479 for a total pressure of 88 kPa for air. Data From Repeat Prob. 1479: Air enters a 40-cm-diameter cooling section at 1 atm, 32C, and 70 percent relative humidity at 120 m/min. The...
-
At 0 K, the entropy of carbon monoxide crystal is not zero but has a value of 4.2 J/K ? mol, called the residual entropy. According to the third law of thermodynamics, this means that the crystal...
-
A system has poles at \(0.01 \mathrm{~Hz}, 1 \mathrm{~Hz}\) and \(80 \mathrm{~Hz}\); zeros at \(5 \mathrm{~Hz}, 100 \mathrm{~Hz}\) and \(200 \mathrm{~Hz}\). The approximate phase of the system...
-
The input-output transfer function of a plant \(\mathrm{H}(s)=\frac{100}{s(s+10)^{2}}\). The plant is placed in a unity negative feedback configuration as shown in the figure below. The signal flow...
-
The following transfer functions do not share a common denominator polynomial, but they may be made to do so by multiplying their numerators and denominators by appropriate factors. Obtain simulation...
-
The forward path transfer function of a unity feedback system is given by \(\mathrm{G}(s)=\frac{1}{(1+s)^{2}}\). What is the phase margin for this system? (a) \(-\pi \operatorname{rad}\) (b) \(0...
-
Consider the following techniques. 1. Bode plot 2. Nyquist plot 3. Nichol's chart 4. Routh-Hurwitz criterion Which of these techniques are used to determine relative stability of a closed loop linear...
-
(a) Calculate the momentum of a 2000-kg elephant charging a hunter at a speed of 7.50 m/s. (b) Compare the elephant's momentum with the momentum of a 0.0400-kg tranquilizer dart fired at a speed of...
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
An ideal-gas mixture consists of 2 kmol of N2 and 6 kmol of CO2. The mass fraction of CO2 in the mixture is (a) 0.175 (b) 0.250 (c) 0.500 (d) 0.750 (e) 0.875
-
An ideal-gas mixture consists of 2 kmol of N2 and 4 kmol of CO2. The apparent gas constant of the mixture is (a) 0.215kJ/kgK (b) 0.225kJ/kgK (c) 0.243kJ/kgK (d) 0.875kJ/kgK (e) 1.24kJ/kgK
-
A rigid tank is divided into two compartments by a partition. One compartment contains 3 kmol of N2 at 400 kPa and the other compartment contains 7 kmol of CO2 at 200 kPa. Now the partition is...
-
Mark has the following utility function: U = ln(w) where w denotes the wealth. Mark has the chance to play in the following lottery: with probability 0.58 the lottery gives $43,750 and with the...
-
DynCorp's FCFF is expected to be $600 million. The firm's interest expense is $80 million. The net debt of the firm decreases by $40 million. The tax rate is 35%. What are the FCFE, the market value...
-
Builtrite has a capital structure of 60% common stock and 40% debt. Bonds being sold will have a $1000 par, 7% coupon rate, a 15 year maturity with a selling price of $1040 and flotation costs of $15...

Study smarter with the SolutionInn App


