Reconsider Prob. 1482. How far will the temperature of the humid air have to be reduced to
Question:
Reconsider Prob. 14–82. How far will the temperature of the humid air have to be reduced to produce the desired dehumidification?
Data From Reconsider Prob. 14–82:
On a summer day in New Orleans, Louisiana, the pressure is 1 atm: the temperature is 32°C; and the relative humidity is 95 percent. This air is to be conditioned to 24°C
and 60 percent relative humidity. Determine the amount of cooling, in kJ, required and water removed, in kg, per 1000 m3 of dry air processed at the entrance to the system.
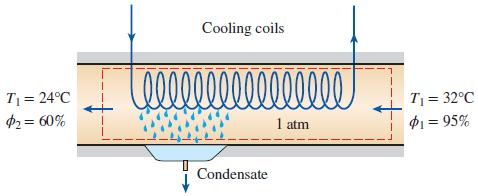
Cooling coils 0000000 T = 32°C P1 = 95% T = 24°C 2= 60% 1 atm Condensate
Step by Step Answer:
The humid air of the previous problem is reconsidered The exit temperature of the air to pr...View the full answer



Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Related Video
In this video, the concept of pressure has been explained. The bottle is filled up with water, remove the pipe, and now you can see the clear flow of water from the holes due to liquid pressure.You can see that the second layer is holding the weight of the first layer and the third layer holding the weight of both upper layers, that’s why liquid flows out wider from the third as compared to the first two.At the first hole, water doesn’t roll along the walls; it flows out at an angle.At the second hole, you’ll see that the water flows out at a wider angle as compared to the first hole.At the third hole, we can see that the flow of water is much wider/greater than the other holes.This proves that the pressure at the bottom of the bottle is much more than the pressure at the top layers. P = ????gh
Students also viewed these Engineering questions
-
On a summer day in New Orleans, Louisiana, the pressure is 1 atm: the temperature is 32C; and the relative humidity is 95 percent. This air is to be conditioned to 24C and 60 percent relative...
-
If the system of Prob. 14-122E is operated as an adiabatic system and the air produced by this system has a relative humidity of 70 percent, what is the temperature of the air produced? Prob. 14-122E...
-
A power company located in southern Alabama wants to predict the peak power load (i.e., Y, the maximum amount of power that must be generated each day to meet demand) as a function of the daily high...
-
Nancy has active modified adjusted gross income before passive losses of $75,000. She has a loss of $5,000 on a rental property she actively manages. How much of the loss is she allowed to take...
-
In a recent year, customers of Victoria Fridge and Stove (which is open 7 days a week) returned 146 appliances. If the frequencies of returns on different days conform to a Poisson distribution, they...
-
Balance sheets for P Company and S Company on August 1, 2019, are as follows: Required: Prepare a workpaper for a consolidated balance sheet for P Company and its subsidiary on August 1, 2019, taking...
-
A sketch of the arithmetic mean radius blade sections of an axial-flow water turbine stage is shown in Fig. P5.90. The rotor speed is \(1000 \mathrm{rpm}\). (a) Sketch and label velocity triangles...
-
On December 31, 2017, Dow Steel Corporation had 600,000 shares of common stock and 300,000 shares of 8%, noncumulative, nonconvertible preferred stock issued and outstanding. Dow issued a 4% common...
-
Submit an annotated outline that includes thematic headings you have drawn from the literature on your topic. The outline must include at least four of the peer-reviewed research articles you found. ...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Atmospheric air from the inside of an automobile enters the evaporator section of the air conditioner at 1 atm, 27C, and 50 percent relative humidity. The air returns to the automobile at 10C and 90...
-
Repeat Prob. 1479 for a total pressure of 88 kPa for air. Data From Repeat Prob. 1479: Air enters a 40-cm-diameter cooling section at 1 atm, 32C, and 70 percent relative humidity at 120 m/min. The...
-
A steel tank of cross-sectional area 3 m 2 and height 16 m weighs 10 000 kg and is open at the top, as shown in Fig. P1.59.Wewant to float it in the ocean so that it is positioned 10 m straight down...
-
The \(\mathrm{dB}\) (Bode plot) of transfer function \(\mathrm{G}(\mathrm{s})\) is shown in figure below. Now, consider the following statements. I. \(\mathrm{G}(s)\) has corner frequencies at...
-
Find decoupled state equations for the system described as \[ \begin{aligned} {\left[\begin{array}{l} \dot{x}_{1} \\ \dot{x}_{2} \end{array}ight] } & =\left[\begin{array}{rr} 0 & 1 \\ -6 & -5...
-
Construct phase variable form simulation diagram for the following transfer functions and develop state space model in matrix form (a) \(\mathrm{T}(s)=\frac{\mathrm{Y}(s)}{\mathrm{U}(s)}=\frac{10...
-
The SISO system with repeated characteristic roots is described by transfer function \[ \mathrm{T}(s)=\frac{\mathrm{Y}(s)}{\mathrm{U}(s)}=\frac{7 s^{3}}{(s+2)^{2}(s+6)^{2}} \] Find state space model...
-
For the system described by the state equation \[ \dot{x}=\left[\begin{array}{ccc} 0 & 1 & 0 \\ 0 & 0 & 1 \\ 0.5 & 1 & 2 \end{array}ight] x+\left[\begin{array}{l} 0 \\ 0 \\ 1 \end{array}ight] u \] if...
-
A person slaps her leg with her hand, bringing her hand to rest in 2.50 milliseconds from an initial speed of 4.00 m/s. (a) What is the average force exerted on the leg, taking the effective mass of...
-
Design an experiment to demonstrate that RNA transcripts are synthesized in the nucleus of eukaryotes and are subsequently transported to the cytoplasm.
-
Repeat Prob. 13-9 by replacing N2 by O2. In Prob. 13-9 A gas mixture has the following composition on a mole basis: 60 percent N2 and 40 percent CO2. Determine the gravimetric analysis of the...
-
Using Amagat's law, show that for a real-gas mixture of k gases, where Z is the compressibility factor. z,- , =D1 Z. i=1
-
An ideal-gas mixture whose apparent molar mass is 20 kg/kmol consists of N2 and three other gases. If the mole fraction of nitrogen is 0.55, its mass fraction is (a) 0.15 (b) 0.23 (c) 0.39 (d) 0.55...
-
A project that is expected to generate annual after-tax cash flows of $2,200 for each of the next 20 years. The project's cost of capital is 8.5 percent and it has an IRR of 9.058 percent. What is...
-
Why are unpaid accruals a "temporary" source of cash? Please give some details and examples.
-
Gabbie has to decide between the following two choices: Option 1: Get $320 with probability 35.97%, and $84.15 with the complement probability. Option 2: Get $94.95 with 100% probability. Her utility...

Study smarter with the SolutionInn App


