A mixture of products of combustion contains the following constituents at 2000 K: 3 kmol of CO
Question:
A mixture of products of combustion contains the following constituents at 2000 K: 3 kmol of CO2, 4 kmol of H2O, and 18.8 kmol of N2. Determine the following quantities:
A. The mole fraction of each constituent in the mixture.B. The apparent molecular weight of the mixture.C. The apparent constant-volume specific heat using a mass-basis (cv,mix in kJ/kg·K) and molar-basis (cv,mixin kJ/kmol·K).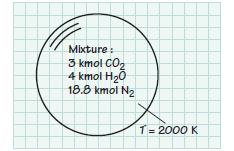
Transcribed Image Text:
Mixture: 3 kmol CO₂ 4 kmol H₂0 18.8 kmol N₂ T= 2000 K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
To solve this problem we need to use the ideal gas law and the definition of mole fraction and appar...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
The combustion of a hydrocarbon fuel with air results in a mixture of products of combustion having the composition on a volume basis as follows: 4.89 percent carbon dioxide, 6.50 percent water...
-
A mixture of ideal gases contains 0.5 kmol of CO 2 , 2 kmol of O 2 , and 7 kmol of N 2 at 700 K. Determine the following quantities: A. The mole fraction of each constituent in the mixture. B. The...
-
The hydroboration-oxidation of internal alkynes produces ketones. (a) When hydroboration-oxidation is applied to but-2-yne, a single pure product is obtained. Determine the structure of this product,...
-
A research laboratory receives delivery of a new custom-made cylindrical thin-wall pressure vessel. The pressure vessel has inner radius R = 300 mm, and wall thickness t = 10 mm. The vessel is made...
-
Review the calculations for The Diamond Ridge Golf Course example discussed in the chapter. 1. Based on these calculations, is there any good-will value? If so, how much? 2. How did you make this...
-
Rather than spending $100 today on paint today, you decide to save the money until next year, at which point you will use it to paint your room. If a can of paint costs $10 today, how many cans will...
-
Refer to the information in Exercise 16-6. Assume that Fields uses the FIFO method of process costing. 1. Calculate the equivalent units of production for the forming department. 2. Calculate the...
-
Write Company has a maximum capacity of 200,000 units per year. Variable manufacturing costs are $12 per unit. Fixed overhead is $600,000 per year. Variable selling and administrative costs are $5...
-
Prove that if M is a dense linear subspace of a separable Hilbert space H, then H has an orthonormal basis consisting of elements in M. Does the same result hold for arbitrary dense subsets of H?
-
Exhibit 5.26 presents risk ratios for Walmart for 2014 and 2013. Exhibits 1.19, 1.20, and 1.21 in Chapter 1 present the financial statements for Walmart. REQUIRED a. Compute the values of each of the...
-
An instrument for the analysis of trace hydrocarbons in air, or in the products of combustion, uses a flame ionization detector. The flame in this device is fueled by a mixture of 40% (vol.) hydrogen...
-
Determine the total apparent specific heat at constant volume (c v,mix in kJ/kgK) for a fuelair reactant mixture containing 1 kmol CH 4 , 2.5 kmol O 2 , and 9.4 kmol N 2 at 500 K and 1 atm. What heat...
-
Identify these values from Table 11. 7: a. C2 b. R1 c. n d. E2,3
-
The action of writing off a past-due accounts receivable is not a task not taken lightly. Let's say your business has a customer that is past-due and owes you a total of $10,000. Your company has...
-
Frederick Griffith's seminal experiment demonstrating DNA as the heritable material utilized two strains of S. pneumonae. One strain (S) was pathogenic, and one strain (R) was non-pathogenic. The S...
-
On 30 June 2023, the statement of financial position of Signature Park Ltd showed the following non-current assets after charging depreciation. Building Accumulated depreciation Motor vehicle...
-
Which question arises at the time property, plant, and equipment is derecognized? ' Question content area bottom Part 1 A. Have the criteria for commercial substance been met? B. When should the...
-
Explain which, if any, of the principles or standards Jeremiah's suggestion will violate. Would your answer change if Wedgewood was a publicly traded company? Would your answer change if Wedgewood...
-
The following transactions are July activities of Bills Extreme Bowling, Inc., which operates several bowling centers. For each of the following transactions, complete the spreadsheet, indicating the...
-
We all experience emotions, but some people disguise their true feelings better than others. Do you think this is a helpful or harmful thing to do? Under what conditions do you think it would be most...
-
You have worked for a petroleum company producing automotive fuels for several years, and in your spare time at home you have developed a new fuel composition that has a higher heating value than...
-
As a production engineer for a large chemical company, you need to find a new supplier for a specific commodity. Since this contract is substantial, the salespeople with whom you meet are naturally...
-
It is December 1928 and your name is Thomas Midgley, Jr.21 You have just invented a new miracle refrigerant composed of chlorinated fluorocarbons (CFCs) that will make your company a lot of money. At...
-
What is the technique of Sharding used by NoSql databases like MongoDb?
-
My Friend intends to develop a financial-related application which is planning to achieve the reliable transactions. He plans to explore the application in the distributed database management system....
-
Many of the NoSQL systems that support large-scale parallel execution guarantee "eventual consistency" rather than the ACID properties. What does ACID mean? What does "eventual consistency" mean?...

Study smarter with the SolutionInn App


