A rigid tank contains 5 kg of O 2 , 8 kg of N 2 , and
Question:
A rigid tank contains 5 kg of O2, 8 kg of N2, and 10 kg of CO2 at 100 kPa and 1000 K. Assume that the mixture behaves as an ideal gas. Determine
(a) The mass fraction of each component,
(b) The mole fraction of each component,
(c) The partial pressure of each component,
(d) The average molar mass (apparent molecular weight) and the gas constant of the mixture,
(e) The volume of the mixture in m3.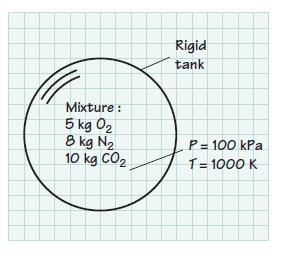
Transcribed Image Text:
Mixture : 5 kg 0₂ 8 kg N₂ 10 kg CO₂ Rigid tank P = 100 kPa T = 1000 K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Given data Mass of O2 5 kg Mass of N2 8 kg Mass of CO2 10 kg Total mass of mixture 23 kg Pressure P ...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
A rigid tank contains 5 kg of saturated vapor steam at 100C. The steam is cooled to the ambient temperature of 25C. (a) Sketch the process with respect to the saturation lines on a T-v diagram. (b)...
-
A rigid tank contains 5 kg of ethylene at 3 MPa, 30C. It is cooled until the ethylene reaches the saturated vapor curve. What is the final temperature?
-
A rigid tank contains 5 kg of ethylene at 3 MPa, 30C. It is cooled until the ethylene reaches the saturated vapor curve. What is the final temperature?
-
9. A molybdenum-vanadium alloy of composition 50wt%Mo - 50wt%V is slowly cooled from a temperature of 2600C to 1800C. Determine: a) At what temperature does the first solid phase form? b) What is the...
-
Barry Means and his spouse Mary want to transfer their one- third (1/ 3) stock interest in The Diamond Ridge Golf Course to a trust for the benefit of their two children. The couples CPA has advised...
-
Solve Prob. 3 when E = E 0 sin t and R, L, E 0 , and ? are arbitrary. Sketch a typical solution. Data from Prob. 3 Model the RL-circuit in Fig. 66. Find a general solution when R, L, E are any...
-
What response the opponent should make to a summary judgment motion?
-
Vixeron Company reported the following on the company's income statement for 2012 and 2011: a. Determine the number of times interest charges were earned for 2011 and 2012. Round to one decimal...
-
Case 1 - Omega Films LTD. Prepare a case analysis being sure to cover the following: 1. What types of transactions are realized between Mrs. Papp and Mr. Holm in the course of negotiations? 2. What...
-
A furniture outlet has made the following wholesale purchases of a new kitchen chairs 18 chairs at $55, 19 chairs at $60, and 12 chairs at $55, An inventory taken last week indicates that 33 chairs...
-
A 3-ft 3 rigid vessel contains a 5050 mixture of N 2 and CO (by volume). Determine the mass of each component for T = 65 F and P = 30 psia.
-
An instrument for the analysis of trace hydrocarbons in air, or in the products of combustion, uses a flame ionization detector. The flame in this device is fueled by a mixture of 40% (vol.) hydrogen...
-
In flowcharts, the decision symbol is a ____________. a. Parallelogram b. Rectangle c. Lozenge d. Diamond
-
We want to increase the temperature of the air contained in a closed bottle. The initial temperature and pressure of the air inside the bottle are 300 K and 202,650 Pa absolute, respectively. The...
-
Tel Tec has just announced that their next dividend will be $2.25. In the announcement, management has projected that earnings will grow at 10 percent per year for the following 3 years, then slow to...
-
Investigate the molecular basis of autophagy, a highly conserved cellular process involved in the degradation and recycling of cellular components, and discuss its multifaceted roles in cellular...
-
I am IDENTITY AWARENESS ACTIVITY I am I am I am I am I am I am I am I am I am I am 1. How did you learn about this identity? When you learned this, how did you feel? 2. What are the advantages and...
-
What is FTX? How does it operate? What factors triggered its bankruptcy? What are the implications for cryptocurrencies at large?
-
A storage system serving an electronics assembly plant has three storage carousels, each with its own manually operated pick and deposit station. The pick and deposit time is 0.30 min. Each carousel...
-
7. FALSE DILEMMA 8. GANDWAGON Definition: Fallacy example: How to revise argument: Definition: Fallacy example: How to revise argument:
-
Repeat exercise 19 for the combustion of coal. For simplicity assume coal is just carbon, C, with a HV of 32,800 kJ/kg. Use engineering considerations to give insight on the global warming issue....
-
Given the heat of formation of liquid methanol, CH 3 OH(l), is 238,000 kJ/kmol, what is its heat of combustion in kJ/kg?
-
In 1800 the main fuel used in the United States was wood. (Assume wood is cellulose, whose representative repeating formula is C 6 H 12 O 6 .) In 1900, the main fuel used was coal (assume coal in...
-
This discussion supports the following objective: Describe the skill sets of health care managers. Explain the relationship between employee motivation and employee engagement. Share a resource that...
-
1. Define the equity method and equity income. 2. What theoretical problems can the components of the equity method identify? 3. Provide specific examples (minimum of two examples) to back up your...
-
I use DBeaver to do the question. In DBeaver, you can choose the Microsoft SQL server for a new database connection, and type that information in page 1 to login the AdventureWorks2008R2 database. ...

Study smarter with the SolutionInn App


