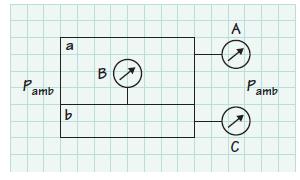
The accompanying sketch shows a compartment divided into two sections a and b. The ambient barometric pressure
Question:
The accompanying sketch shows a compartment divided into two sections a and b. The ambient barometric pressure reading is 30.0 inches of mercury (absolute). Gage C reads 620,528 Pa and gage B reads 275,790 Pa. Determine the reading of gage A and convert this reading to an absolute value.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Conversion factors given in text inside front cover st...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
How many years are required for $660 to grow to $1,254 at 18 simple interest Determine the formula and values that should be used to solve this problem if r represents the annual interest rate in...
-
The sketch for Problem 2.22 shows a compartment divided into two sections a and b. The ambient barometric pressure reading is 30.0 inches of mercury (absolute). Gage C reads 615 kPa and gage B reads...
-
The sketch for Problem 2.22 shows a compartment divided into two sections a and b. The ambient barometric pressure reading is 29.0 inches of mercury (absolute). Gage A reads 435 kPa and gage B reads...
-
In the conversion of a-ketoglutarate to glucose, which of the following compounds are not obligatory intermediates in this pathway? * Oxaloacetate O Fructose 1,6 bisphosphate O Malate O 1,3...
-
Required a. Identify which of the following accounts are temporary (will be closed to Retained Earnings at the end of the year) and which are permanent: (1) Other Operating Expenses (2) Utilities...
-
The tensile strength and number-average molecular weight for two poly (methyl methacrylate) materials are as follows: Tensile Strength (MPa) ____________ Number-Average Molecular Weight (g/mol) 50...
-
Which of the following terms describe the types of adjusting entries? a. Deferrals and depreciation b. Expenses and revenues C. Deferrals and accruals d. Prepaid expenses and prepaid revenues
-
Sand from a stationary hopper falls onto a moving conveyor belt at the rate of 5.00 kg/s as in Figure P9.72 The conveyor belt is supported by frictionless rollers and moves at a constant speed of...
-
1) Draw the graph of the following relation and state the domain and range. 5, =] x+3, -2
-
Students should read the Harvard Business School case study entitled 'Investments: Delineating an Efficient Portfolio' and answer the following questions. Students should note that the risk of the...
-
The pressure in a partially evacuated enclosure is 26.8 inches of mercury when the local barometer reads 29.5 inches of mercury. Determine the absolute pressure in units of inches of mercury, psia,...
-
The sketch for Problem 2.22 shows a compartment divided into two sections a and b. The ambient barometric pressure reading is 28.0 inches of mercury (absolute). Gage A reads 5 kPa and gage B reads...
-
What are the self-employment tax rates for 2018? Explain how the tax is computed.
-
Normal profit is best described as: A. zero economic profit. B. total revenue minus all explicit costs. C. the sum of accounting profit plus economic profit.
-
Which of the following statements best explains why indifference curves are generally convex as viewed from the origin? A. The assumption of nonsatiation results in convex indifference curves. B. The...
-
The component least likely to be included in a measurement of gross domestic product (GDP) is: A. the value of owner-occupied rent. B. the annual salary of a local police officer. C. environmental...
-
A child indicates that she prefers going to the zoo over the park and prefers going to the beach over the zoo. When given the choice between the park and the beach, she chooses the park. Which of the...
-
In an industry comprised of three companies that are small-scale manufacturers of an easily replicable product unprotected by brand recognition or patents, the most representative model of company...
-
Evaluate the appropriateness of the McCafé line of coffees and the fruit-and-maple oatmeal to McDonald's overall merchandising strategy.
-
Which one of the following anhydrous chloride is not obtained on direct heating of its hydrated chloride? (A) BaCl2 (B) CaClz (C) MgCl2 (D) SrCl2
-
A 20.0-mL solution of 0.005 00 M Sn 2+ in 1 M HCl was titrated with 0.020 0 M Ce 4+ to give Sn 4+ and Ce 3+ . Calculate the potential (versus S.C.E.) at the following volumes of Ce 4+ : 0.100, 1.00,...
-
Would indigo tetrasulfonate be a suitable redox indicator for the titration of Fe(CN) 6 4- with Tl 3+ in 1 M HCl? The potential at the equivalence point must be between the potentials for each redox...
-
Compute the titration curve for Demonstration 15-1, in which 400.0 mL of 3.75 mM Fe 2+ are titrated with 20.0 mM MnO 4 - at a fixed pH of 0.00 in 1 M H 2 SO 4 . Calculate the potential versus S.C.E....
-
What is Effective / Economic incidence and Legal incidence in Tax ?
-
A firm has a capital structure which is composed of 50% debt and 50% equity. If the debt rate is 9.77% and the rate of return on equity is 12.65%, calculate the cost of capital.
-
Kirksville Corp. is considering a new assembly line to replace the existing assembly line. The assembly line would require to use a parcel of land that cost $630,000 three years ago. But the land can...

Study smarter with the SolutionInn App


