What is the temperature or quality of H 2 O in the following states? A:
Question:
What is the temperature or quality of H2O in the following states?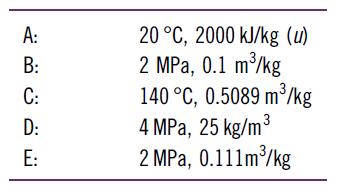
Transcribed Image Text:
A: ننا ت ت ق ة B: C: D: E: 20 °C, 2000 kJ/kg (u) 2 MPa, 0.1 m3/kg 140 °C, 0.5089 m/kg 4 MPa, 25 kg/m 3 2 MPa, 0.111m3/kg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
ANSWER Using the information provided in the image we can determine the temperature and quality of w...View the full answer

Answered By

Churchil Mino
I have been a tutor for 2 years and have experience working with students of all ages and abilities. I am comfortable working with students one-on-one or in small groups, and am able to adapt my teaching style to meet the needs of each individual. I am patient and supportive, and my goal is to help my students succeed.
I have a strong background in math and science, and have tutored students in these subjects at all levels, from elementary school to college. I have also helped students prepare for standardized tests such as the SAT and ACT. In addition to academic tutoring, I have also worked as a swim coach and a camp counselor, and have experience working with children with special needs.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
What is the temperature that corresponds to 1.8-TeV collisions at the Fermilab Collider? To what era in cosmological history does this correspond?
-
What is the temperature inside an ideal refrigerator freezer that operates with a COP = 7.0 in a 22C room?
-
The hottest temperature ever recorded on the surface of the earth was 136F in Libya in 1922. What is the temperature in degrees Celsius and in kelvins?
-
As manager of a local pizza parlor, you want to develop a balanced scorecard so you can more effectively monitor the restaurants performance. Required a. Propose at least two goals for each...
-
Zycor Corporation obtains most of its funding internally. Assume that the stock has a beta of 1.2, the riskless rate is 6.5%, and the market risk premium is 6%. a. Estimate the cost of internal...
-
Use conditional proof and the eighteen rules of inference to derive the conclusions of the following symbolized arguments. Having done so, attempt to derive the conclusions without using conditional...
-
Journalize the following transactions for Antique Furniture, Inc., that occurred during the month of April. Antique Furnitures cost of inventory is 70% of the sales price. Apr 3 Sold $2,100 of...
-
At the end of 20X5, Singh Inc. has four inventory items, two of which management believes should be written down. The cost and estimated NRVs of the items are as follows: Required: 1. Determine the...
-
a) A company wants to expand to 2 new locations in New York. Company executives believe that this would initially cost $20 Millions to expand. The yearly FCF will begin at $4 milion in year 1 and...
-
An automatic machine produces (thousands of) units of a product per day. As increases, the proportion of defectives, p, goes up according to the following probability density function Each...
-
Steam is condensing in the shell of a heat exchanger at 305 K under steady conditions. The volume of the shell is 2.75m 3 . Determine the mass of the liquid in the shell if the specific enthalpy of...
-
How to Use the NIST Software. Given the following property data for H 2 O, designate the region, line, or point in Tv or Pv space (i.e., compressed liquid, liquidvapor mixture, superheated vapor,...
-
What benefits are there in replacing all existing UK GAAP standards with one overall standard (draft FRS 102)?
-
What are the factors in the Fama-French model?
-
What is short-term momentum? What are long-term reversals?
-
What violations of the EMH have been demonstrated?
-
What does the term attainable set mean?
-
What are the key assumptions of the CAPM?
-
Discuss the pros and cons of evaluating a retailer's success using same store sales growth data.
-
KD Insurance Company specializes in term life insurance contracts. Cash collection experience shows that 20 percent of billed premiums are collected in the month before they are due, 60 percent are...
-
A natural-gas-fired electrical power plant produces an output of 750 MW. By using a typical efficiency from Table 7.6 and neglecting the small amount of power drawn by the pump, calculate the rates...
-
Suppose that the new type of computer power supply described in Problem P7.31 costs an additional $5. (a) At the cost of 12 per kW ? h, after what period of time would the cost savings in electricity...
-
For the plant in Problem P7.33, 25,000 gal/s of water flow in the river adjacent to the power plant. The river is used as the source of cooling water for the condenser. Considering the heat...
-
Explain completely Why are customer perceptions one of the most critical aspects of exemplary customer service?
-
Kmeciak Corp. incurs unit costs of $6.20 ($5.10 variable and $1.10 fixed) in making a gear for its finished product. A supplier offers to make 8,300 of the assembly part at $5.30 per unit. If the...
-
Sales for 2018 were $455,150 and EBITDA was 15% of sales. Furthermore, depreciation and amortization were 11% of net fixed assets, interest was $8,575, the corporate tax rate was 40%, and Laiho pays...

Study smarter with the SolutionInn App


