An ideal gas is characterised by the relation pV = NRT where p is the pressure of
Question:
An ideal gas is characterised by the relation pV = NRT where p is the pressure of the gas, V is the volume, T is the temperature, N is the number of moles of gas and R is a constant.a) Calculate the differential dp (T, V)
b) Calculate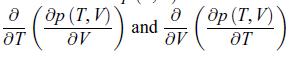
Transcribed Image Text:
8 ( 8p ( T, V ) ) OT 8Ꮴ and 8 ( Ꭷp ( T, V ) ᎥᏙ ᎧᎢ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a To calculate the differential dp T V we first need to find the partial derivatives of p with respe...View the full answer

Answered By

Anik Kumar Bosu
I am expert in Maths, Physics, Chemistry and Biology. I also teach student from 3 years. Students becomes satisfied by my teaching. So, do not worry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Principles Of Thermodynamics
ISBN: 9781108426091
1st Edition
Authors: Jean-Philippe Ansermet, Sylvain D. Brechet
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas is characterised by the relation pV = NR T as in 1.2 where the pressure p (T, V) is a function of T and V, the temperature T (p, V) is a function of p and V and the volume (T, p) is a...
-
A gas is characterised by the enthalpy H(S, p) = C p T, where C p is a constant (called heat capacity and defined in 5.2), and by pV = NR T, where p is its pressure, V its volume, T its temperature...
-
Thermal Expansion of an Ideal Gas (a) The pressure p, volume V, number of moles n, and Kelvin temperature T of an ideal gas are related by the equation p V = nRT, where R is a constant. Prove that...
-
What are the "two main lines" of distortion of the view of Marx and what does Lenin think the proper view?
-
Record the following annuity and life income activities of Private University: 1. On July 1, 2010, R. W. Fields, emeritus professor of accounting, moved out of the state. Fields donated to the...
-
A voluntary health and welfare organization summarizes its expenses by function, as follows: Education........ $20,400 Fund-raising...... 11,400 Management and general.. 5,500 Public health.........
-
According to Boyle's law for gases (a) \(V \propto T\) while \(P=\) constant (b) \(V \propto 1 / P\) while \(T=\) constant (c) \(V \propto 1 / T\) while \(P=\) constant (d) \(V \propto P\) while...
-
Starbucks is a resounding restaurant success story. Beginning with its first coffee house in 1971, Starbucks has grown to more than 11,000 U.S. locations. Opening up its first international outlet in...
-
Artifact 1: Create a resource for colleagues: Codes of Ethics and Highlights Explained Design a visually appealing resource to share with your colleagues that explains highlights of NEA's Code of...
-
(a) Show that the transfer function of two systems in parallel, as shown in Fig. P1.1-l(a), is equal to the sum of the transfer functions. (b) Show that the transfer function of two systems in series...
-
A basin contains Ns (t) moles of salt dissolved in Nw (t) moles of water. The basin receives fresh water at a constant rate in w. This water is assumed to be thoroughly mixed in the basin so that the...
-
Two of the Massieu functions are functions of the following state variables: 1. 2. The Massieu functions are obtained by performing Legendre transformations of the state function entropy S (U, V)...
-
Reconsider Prob. 1199. Using EES (or other) software, investigate the effects of the inlet temperature of hot water and the heat transfer coefficient on the rate of heat transfer and the surface...
-
In your organization, it is traditional to give everyone involved in the project a printed copy of the design specifications after they have been approved. It costs more, but management feels this is...
-
Bittoto Ltd has four different intangible assets at the end of 2019. Facts concerning each are as follows: 1. franchise. On 2 January 2019, Bittoto Ltd purchased a franchise to distribute a new...
-
You work for a consulting company that has been hired to do the systems design portion of the project. The systems analysis portion was done by another consulting company. During systems design, you...
-
You are a systems analyst who has been working for several years in the IT shop of a cabinet manufacturing company. The company is known for the quality of its products and for being an industry...
-
In the first question, you were asked to look at a variety of approaches to systems design, including prototyping. In the past several years, the number of prototyping methodologies and application...
-
Consider the probability distribution of a random variable x. Is the expected value of the distribution necessarily one of the possible values of x? Explain or give an example.
-
Ex. (17): the vector field F = x i-zj + yz k is defined over the volume of the cuboid given by 0x a,0 y b, 0zc, enclosing the surface S. Evaluate the surface integral ff, F. ds?
-
A solution of NaOH was standardized by titration of a known quantity of the primary standard, potassium hydrogen phthalate The NaOH was then used to find the concentration of an unknown solution of H...
-
Write the names and abbreviations for each of the prefixes from 10 -24 to 10 24 . Which abbreviations are capitalized?
-
What is the formal concentration (expressed as mol/L = M) of NaCl when 32.0 g are dissolved in water and diluted to 0.500 L?
-
W Nov. 8 8 11 11 13 14 16 Natalie cashes her U.S. Savings Bonds and receives $630, which she deposits in her personal bank account. She opens a bank account under the name "Cookie Creations" and...
-
Besides the effect on the tax rates that will apply, filing status also has an impact on the amount of the standard deduction that is allowed. A taxpayer who is unmarried or separated from their...
-
On January 1 of its first month of business, Juan in a Million, Inc., paid $48,000 for four months rent beginning in January. How much will be reported as Rent Expense on its income statement for the...

Study smarter with the SolutionInn App


