An ideal gas is characterised by the relation pV = NR T as in 1.2 where
Question:
An ideal gas is characterised by the relation pV = NR T as in § 1.2 where the pressure p (T, V) is a function of T and V, the temperature T (p, V) is a function of p and V and the volume (T, p) is a function of T and p. Calculate,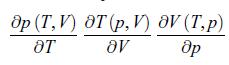
Transcribed Image Text:
др (T, V) OT (P, V) 8V (T,P) OT aV др
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
To find the partial derivatives we will use the relations pV NR T equation 1 p pT V eq...View the full answer

Answered By

Jayka Depante
As a tutor I am very friendly . Since last year I am giving IIT JAM tuition , among 9 students , 7 cracked this exam this year. I will try to teach every topic with practical examples. students are just like friends to me.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Principles Of Thermodynamics
ISBN: 9781108426091
1st Edition
Authors: Jean-Philippe Ansermet, Sylvain D. Brechet
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas is characterised by the relation pV = NRT where p is the pressure of the gas, V is the volume, T is the temperature, N is the number of moles of gas and R is a constant.a) Calculate the...
-
A gas is characterised by the enthalpy H(S, p) = C p T, where C p is a constant (called heat capacity and defined in 5.2), and by pV = NR T, where p is its pressure, V its volume, T its temperature...
-
Using java language and data structure In chapter 1 you learned how to merge two sorted lists and get a third list which is also sorted. Write a method named mergeUnsortedLists in an application,...
-
Record the following capital-related transactions for Private University plant funds: 1. Transfers of $250,000 are received from the current unrestricted fund for the purpose of funding the payment...
-
Explain the difference between donor-imposed conditions and donor-imposed restrictions.
-
The heat capacity at constant pressure \(\left(C_{P} ight)\) is defined as (a) \(C_{P}=\left(\frac{\partial U}{\partial T} ight)_{P}\) (b) \(C_{P}=\left(\frac{\partial Q}{\partial T} ight)_{P}\) (c)...
-
After watching the video on GM in Shanghai, discuss the following: 1. How would you characterize the alliance between GM and SAIC Shanghai? 2. What has driven the GM alliance in Shanghai? 3. How is...
-
Why do you think media sends camera crews and reporters to town-hall meetings? What role do they play? Go to thewww.ottawa.ca and browse the website. Then go towww.renfrew.ca and browse their...
-
Montreal Electronics Company manufactures two large-screen television models: the Nova, which has been produced for 10 years and sells for $900, and the Royal, a new model introduced in early 20x0,...
-
A basin contains Ns (t) moles of salt dissolved in Nw (t) moles of water. The basin receives fresh water at a constant rate in w. This water is assumed to be thoroughly mixed in the basin so that the...
-
Two of the Massieu functions are functions of the following state variables: 1. 2. The Massieu functions are obtained by performing Legendre transformations of the state function entropy S (U, V)...
-
The Ipsos polling organization conducted a national survey in 2019, finding that 295 of the sample of 1,005 American adults had a tattoo. a. Use the Theory-Based Inference applet to determine a 99%...
-
On 1 January 2017, Nicolaidis Ltd purchased two identical new machines at a total cost of $700 000. It was estimated that the machines would have a useful life of 10 years and a residual value of $50...
-
You are designing a data interface screen for a new system that is under development. The purpose of this data interface screen is to enter changes of address submitted by drivers to their states...
-
Over a 5year period, Downton Ltd completed the following transactions affecting noncurrent assets in financial years ending 31 December. The company uses straightline depreciation on all depreciable...
-
You are a systems designer in an organization. One of the other designers on your team has recently retired. Your manager comes to you and asks you to sit in on the interviews to fi nd a replacement....
-
Consider the issues raised by the preceding question and write a one- or two-page policy and procedures memorandum to all systems analysts and designers in your organization regarding prototyping.
-
Consider the probability distribution shown in Problem 3(a). Compute the expected value and the standard deviation of the distribution.
-
Reduction in sales All of the above 29. Belt of an electric motor is broken, it needs a. Corrective maintenance b. Scheduled maintenance c. Preventive maintenance d. Timely maintenance. 30. The...
-
A 48.0 wt% solution of HBr in water has a density of 1.50 g/mL. (a) Find the formal concentration of HBr. (b) What mass of solution contains 36.0 g of HBr? (c) What volume of solution contains 233...
-
A solution contains 12.6 ppm of dissolved Ca(NO 3 ) 2 (which dissociates into Ca 2+ 2NO - 3 ). Find the concentration of NO - 3 in parts per million.
-
Ascorbic acid (vitamin C, page 354) reacts with I - 3 according to the equation Starch is used as an indicator in the reaction. The end point is marked by the appearance of a deep blue starch-iodine...
-
Sweet Catering completed the following selected transactions during May 2024: (Click the icon to view the transactions.) Read the requirements. Requirement 1. Show whether each transaction would be...
-
Warnerwoods Company uses a periodic inventory system. It entered into the following purchases and sales transactions for March. Date March 1 March 5 Activities Beginning inventory Purchase Units...
-
You have already learned that when preparing financial statements, management makes assertions about each account and the relate disclosures in the notes. Auditors then gather sufficient appropriate...

Study smarter with the SolutionInn App


