Calculate the density of gaseous CO 2 at each of the following states:a. 10 bar, 290 K,b.
Question:
Calculate the density of gaseous CO2 at each of the following states:a. 10 bar, 290 K,b. 30 bar, 333 K,c. 100 bar, 410 K,Using the perfect gas model. Compare the values obtained with the values listed in the tables in Appendix A.9. What can you infer?
Data From Appendix A.9
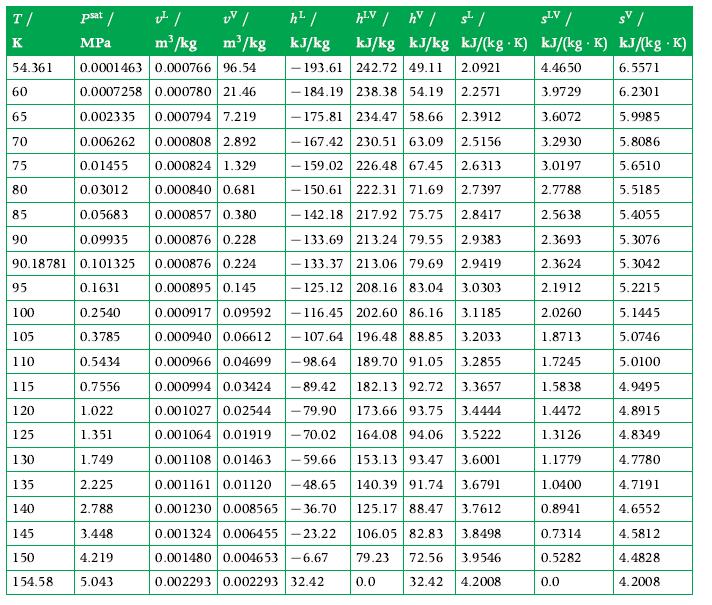
Transcribed Image Text:
T/ K UV/ m³/kg m³/kg 54.361 0.0001463 0.000766 96.54 0.0007258 0.000780 21.46 0.002335 0.000794 7.219 0.000808 2.892 0.000824 1.329 0.006262 0.01455 0.03012 0.000840 0.681 0.05683 0.000857 0.380 0.09935 0.000876 0.228 90.18781 0.101325 0.000876 0.224 0.1631 0.000895 0.145 60 65 70 75 80 85 psat / MPa 90 95 100 0.2540 105 0.3785 110 0.5434 115 0.7556 120 1.022 125 1.351 130 1.749 135 2.225 140 2.788 145 3.448 150 4.219 154.58 5.043 h² / kJ/kg hLV / hv / 5¹/ SLV / SV/ kJ/kg kJ/kg kJ/(kg K) kJ/(kg. K) kJ/(kg .K) 4.4650 6.5571 3.9729 6.2301 3.6072 5.9985 3.2930 5.8086 3.0197 5.6510 2.7788 5.5185 2.5638 5.4055 2.3693 5.3076 2.3624 5.3042 2.1912 5.2215 2.0260 5.1445 1.8713 5.0746 1.7245 5.0100 1.5838 4.9495 1.4472 4.8915 1.3126 4.8349 1.1779 4.7780 1.0400 4.7191 0.8941 4.6552 0.7314 4.5812 0.5282 4.4828 4.2008 193.61 242.72 49.11 2.0921 184.19 238.38 54.19 2.2571 175.81 234.47 58.66 2.3912 - 167.42 230.51 63.09 2.5156 -159.02 226.48 67.45 2.6313 -150.61 222.31 71.69 2.7397 -142.18 217.92 75.75 2.8417 2.9383 -133.69 213.24 79.55 133.37 213.06 79.69 2.9419 -125.12 208.16 83.04 3.0303 0.000917 0.09592 -116.45 202.60 86.16 3.1185 0.000940 0.06612 -107.64 196.48 88.85 3.2033 182.13 92.72 3.3657 0.000966 0.04699 -98.64 189.70 91.05 3.2855 0.000994 0.03424 -89.42 0.001027 0.02544 - 79.90 0.001064 0.01919 - 70.02 173.66 93.75 3.4444 164.08 94.06 3.5222 153.13 93.47 3.6001 0.001108 0.01463 -59.66 0.001161 0.01120 -48.65 3.6791 140.39 91.74 0.001230 0.008565-36.70 125.17 88.47 3.7612 0.001324 0.006455-23.22 0.001480 0.004653-6.67 0.002293 0.002293 32.42 106.05 82.83 3.8498 79.23 72.56 3.9546 0.0 32.42 4.2008 0.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
To calculate the density of gaseous CO2 at each of the given states using the perfect gas model we c...View the full answer

Answered By

Monette Taban
I am currently studying Computer Science Engineering, Due to my interest in programming languages and coding, I am interesetd on Technology so I search about it read about different types of technologies, I think my this habbis will help me to solve problems of students and that is why I am signing as a question answer expert.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
In order to test the applicability of the ideal gas equation of state to calculate the density of saturated steam, compare the specific volume of saturated steam obtained from the steam table with...
-
(a) Calculate the density of the atmosphere at the surface of Mars (where the pressure is 650 Pa and the temperature is typically 253 K, with a CO 2 atmosphere), Venus (with an average temperature of...
-
Calculate the density of states g (E) for the free-electron model of a metal if E = 7.0eV and V = 1.0 cm3. Express your answer in units of states per electron volt.
-
Let be an arbitrary operation in Problems 5259. Describe the operation for each problem. 5038; 70 2= 9; 901 = 10; 8 0 2 = 10; -
-
If a potential investments internal rate of return is above the companys hurdle rate, should the investment be made?
-
Figure is a portion of the titanium-copper phase diagram for which only single-phase regions are labeled. Specify all temperature-composition points at which eutectics, eutectoids, peritectics, and...
-
What characteristics do all religions have in common?
-
On October 31, 20Y5, the balances of the accounts appearing in the ledger of Quality Interiors Company, a furniture wholesaler, are as follows: a. Prepare a multiple-step income statement for the...
-
A new English -Language Arts (ELA) teacher is so excited to take all of the ideas she learned during her training and use them in the classroom. After a PLC (Professional Learning Committee) meeting,...
-
You are to develop a simple Binary Search Tree ADT and run it against a test program. Avoid the temptation of finding code online. I am aware of all the available solutions and will be looking...
-
Propane is in a 5 m 3 container as a saturated vapor at 300 K. The sealed tank is then cooled to 260 K by fresh snow. What are the pressure (Pa) and liquid volume in this 260 K state, and how much...
-
Consider each matrix as the augmented matrix of a linear system. State in words the next two elementary row operations that should be performed in the process of solving the system. 1-6 4 2 -7 0 0...
-
Predict whether unemployment will increase or decrease as a result of each of the following monetary policies. If it is unanticipated? What if it is anticipated? a. A reduction in the discount rate...
-
Google has an 80 percent market share in search advertising. It is a monopoly. In the early 20th century, Supreme Court Justice Louis Brandeis said, in a democratic society the existence of large...
-
How does Uniqlos marketing change the firms cost and revenue curves and its economic profit? Does average total cost at the profit-maximizing output increase or decrease? Uniqlos marketing seeks to...
-
In 2015, an outbreak of Avian Flu decreased the quantity of eggs produced by 18 percent. A shortage of eggs was avoided by a rise in their wholesale price from $1.34 to $2.40 per dozen. a. If the...
-
List and comment on the advantages and disadvantages of both full and partial storage systems.
-
Mitsubishi to Close U.S. Auto Plant Mitsubishi Motors will close its production plant in Normal, Illinois. Dwindling sales in North America have brought a loss in the region of $200 million in the...
-
Draw the silicone that can be made from this monomer: Si Si
-
Consider the sections of two circuits illustrated above. Select True or False for all statements.After connecting a and b to a battery, the voltage across R1 always equals the voltage across R2.Rcd...
-
A world-class runner can run half a mile in a time of 1 min and 45 s. What is the runners average speed in m/s?
-
One U.S. gallon is equivalent to 0.1337 ft 3 , 1 ft is equivalent to 0.3048 m, and 1000 L are equivalent to 1 m 3 . By using those definitions, determine the conversion factor between gallons and...
-
A passenger automobile is advertised as having a fuel economy rating of 29 mi/gal for highway driving. Express the rating in the units of km/L.
-
Determine if the following process is allowed by the strong interaction. If it is not allowed, determine which conservation law forbids the process.
-
On November 1 2 , XYZ Corporation declared a total cash dividend of $ 4 5 , 0 0 0 for stockholders of record on November 2 0 and payable on December 1 . ( a ) Journalize the entries required on...
-
How does the motif of mirrors and reflections serve to explore themes of identity and self-perception in Gothic literature, and how do these themes manifest in the psychological evolution of key...

Study smarter with the SolutionInn App


