Propane is in a 5 m 3 container as a saturated vapor at 300 K. The sealed
Question:
Propane is in a 5 m3 container as a saturated vapor at 300 K. The sealed tank is then cooled to 260 K by fresh snow. What are the pressure (Pa) and liquid volume in this 260 K state, and how much energy was transferred as heat from the propane? Use the tables in Appendix A.6 for data.
Data From Appendix A.6
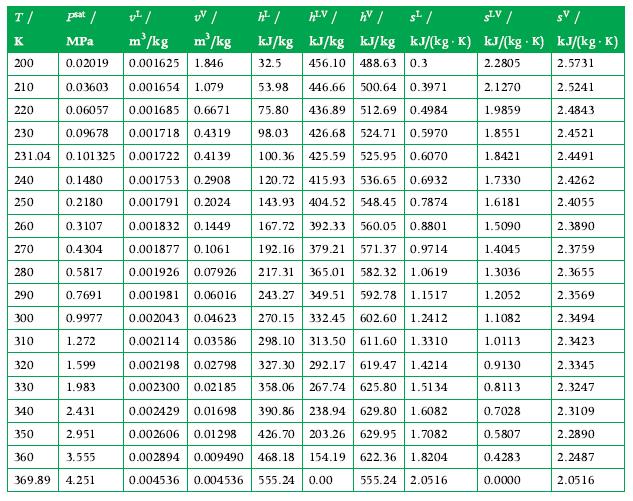
Transcribed Image Text:
T/ K 200 210 0.03603 0.001654 1.079 220 0.06057 0.001685 0.6671 230 0.09678 0.001718 0.4319 231.04 0.101325 0.001722 0.41 39 0.1480 0.001753 0.2908 0.001791 0.2024 0.2180 0.3107 0.001832 0.1449 0.4304 0.001877 0.1061 0.5817 0.001926 0.07926 0.001981 0.06016 0.7691 0.9977 0.002043 0.04623 1.272 0.002114 0.03586 240 250 260 270 280 290 300 310 psat / v²/ UV/ MPa m³/kg m³/kg 0.02019 0.001625 1.846 320 330 1.599 1.983 2.431 2.951 3.555 369.89 4.251 340 350 360 h² / kJ/kg 32.5 HLV / AV / kJ/kg kJ/kg 456.10 488.63 0.3 53.98 446.66 500.64 0.3971 75.80 436.89 512.69 0.4984 98.03 426.68 524.71 0.5970 100.36 425.59 525.95 0.6070 s²/ SLV/ sv/ . kJ/(kg. K) kJ/(kg K) kJ/(kg. K) 2.2805 2.5731 2.5241 2.4843 2.4521 2.4491 120.72 415.93 536.65 0.6932 143.93 404.52 548.45 0.7874 167.72 392.33 560.05 0.8801 192.16 379.21 571.37 0.9714 217.31 365.01 582.32 1.0619 243.27 349.51 592.78 1.1517 270.15 332.45 602.60 1.2412 298.10 313.50 611.60 1.3310 327.30 292.17 619.47 1.4214 358.06 267.74 625.80 1.5134 0.002198 0.02798 0.002300 0.02185 0.002429 0.01698 390.86 238.94 629.80 1.6082 0.002606 0.01298 426.70 203.26 629.95 1.7082 0.002894 0.009490 468.18 154.19 622.36 1.8204 0.004536 0.004536 555.24 0.00 555.24 2.0516 2.1270 1.9859 1.8551 1.8421 1.7330 1.6181 1.5090 1.4045 1.3036 1.2052 1.1082 1.0113 0.9130 0.8113 0.7028 0.5807 0.4283 0.0000 2.4262 2.4055 2.3890 2.3759 2.3655 2.3569 2.3494 2.3423 2.3345 2.3247 2.3109 2.2890 2.2487 2.0516
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To solve this problem we will use the following steps Determine the initial state of propane at 300 K Calculate the final state of propane at 260 K as...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
Values of the specific internal energy of bromine at three conditions are listed here. (a) What reference state was used to generate the listed specific internal energies? (b) Calculate U(kJ/mol) for...
-
The vapor-phase molar volume of a particular compound is reported as 23,000 cm3 mol-1 at 300 K and 1 bar. No other data are available. Without assuming ideal-gas behavior, determine a reasonable...
-
Values of the specific internal energy of bromine at three conditions are listed here. (a) What reference (b) Calculate U (kJ/mol) for a process in which bromine vapor at 300 K is condensed at...
-
Let be an arbitrary operation in Problems 5259. Describe the operation for each problem. 503 = 2; 702= 5; 901 = 8; 8 0 2 = 6;
-
Why does the use of the accelerated depreciation method (instead of straight-line) for income tax reporting increase an investments value?
-
Mr . S . Krishna received a gift of a house in Nov. 2 0 0 7 from Mr . K . Hegde who had purchased it in Nov. 1 9 9 7 for 6 , 3 0 , 0 0 0 . Mr . K . Hegde renovated the house in March 1 9 9 8 at a...
-
The data below pertains to the number of hours a laptop has been charged for and the number of hours of backup provided by the battery. (a) Use the first set of expressions on page 330, involving...
-
Balanced Scorecard or activity-based costing Suppose an organization has not implemented either activity-based costing or a Balanced Scorecard but believes both would be valuable for the...
-
Explore the application of advanced fluid dynamics concepts in aerodynamics and hydrodynamics, analyzing the principles of lift, drag, and thrust, and discussing the design and optimization of...
-
Hemming Company reported the following current-year purchases and sales for its only product. Date Activities January 11 Beginning inventory. January 10 Sales March 14 March 151 July 30 October 5...
-
A constant volume vessel of 100 liters holds 1 kg of water at an initial pressure of 30 bar. The container is then cooled to 305 K. Determine the initial temperature of the system as well as the...
-
Calculate the density of gaseous CO 2 at each of the following states:a. 10 bar, 290 K,b. 30 bar, 333 K,c. 100 bar, 410 K,Using the perfect gas model. Compare the values obtained with the values...
-
What steps should a manager of a limited-service hotel offering complimentary alcoholic beverages during a Manager's Reception take to help ensure that guests do not over-consume these beverages?
-
A contract requires end-of-month payments of $245 for another 8 1/4 years. What would an investor pay to purchase this contract if she requires a rate of return of 1.8% compounded monthly? (Do not...
-
Assume a corporation' s bond has 10 years remaining until maturity . The bond has a $1,000 face value and a 6% coupon rate each year. Other investments meeting the same criteria must provide annual...
-
1. What is the lower bound for the price of a 3-month European call option on a non-dividend paying stock when the stock price is $100, the strike price is $90, and the risk-free interest rate is 5%...
-
Suppose that many people choose between becoming nurses and becoming occupational therapists. The wages of nurses have risen dramatically since the COVID-19 pandemic. How will this affect the wages...
-
Predict the output of this Java code: public class Driver { public int x = 0; public static int y = 0; public static void main(String[] args) { Driver dl new Driver(); Driver d2 = new Driver();...
-
To motivate individuals to start saving at an early age, financial planners will sometimes present the results of the following type of calculation. How much must a 25-year-old individual invest five...
-
A regular deposit of $100 is made at the beginning of each year for 20 years. Simple interest is calculated at i% per year for the 20 years. At the end of the 20-year period, the total interest in...
-
Consider the motion of the piston discussed in Example 1.61. The piston stroke is the total distance moved by the piston as the crank angle varies from 0 to 180. a. How does the piston stroke depend...
-
a. Use two methods to create the vector x having 100 regularly spaced values starting at 5 and ending at 28. b. Use two methods to create the vector x having a regular spacing of 0.2 starting at 2...
-
The wind power output per unit area swept by the rotor is 2.4 kW/m 2 . Convert this quantity to the dimensions of hp/ft 2 .
-
Evalue ! d2 , 2(2+in) , where c is circle. 12+3:1 F1 by using cauchy's Integral. formula.
-
How do the monetization strategies of social networks, such as targeted advertising and user data exploitation, impact user experience and societal norms ?
-
what ways do social structures and institutional power dynamics perpetuate systemic inequalities across different societies, and how might these structures be effectively challenged or transformed to...

Study smarter with the SolutionInn App


