Calculate the values of specific volume, specific internal energy, and specific enthalpy for water in vaporliquid equilibrium
Question:
Calculate the values of specific volume, specific internal energy, and specific enthalpy for water in vapor–liquid equilibrium at 180°C if its quality is 0.2. Use the tables in Appendix A.3 for data.
Data From Appendix A.3
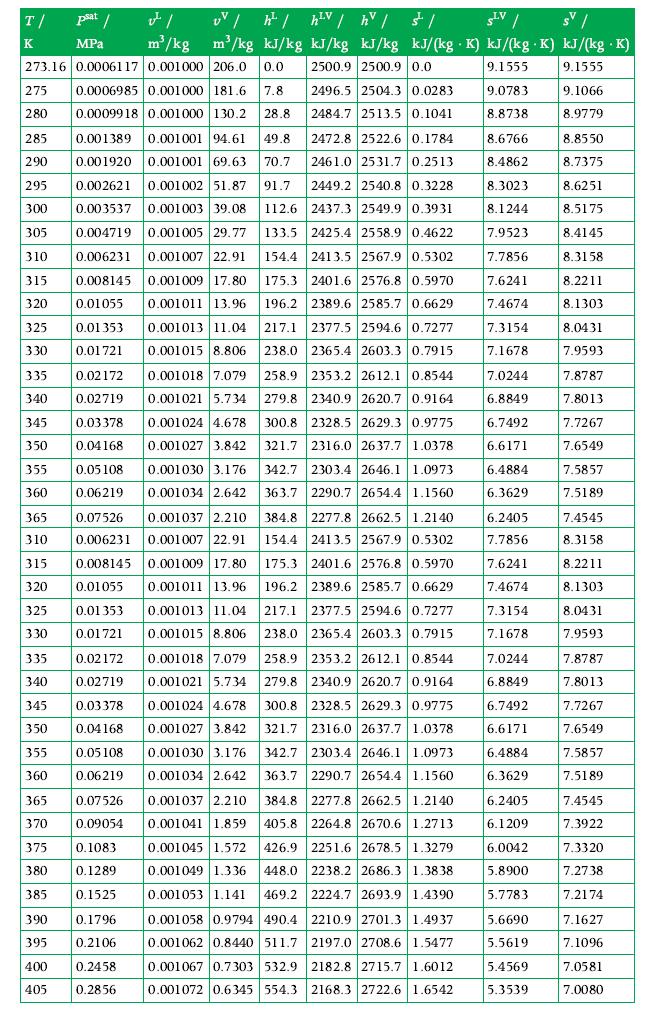
Transcribed Image Text:
T/ psat / MPa K v m³/kg 273.16 0.0006117 0.001000 206.0 0.0 0.0006985 0.001000 181.6 7.8 0.0009918 0.001000 130.2 28.8 335 340 345 350 355 360 UV /h²/h²V / hv / / SLV / sv/ m³/kg kJ/kg kJ/kg kJ/kg kJ/(kg K) kJ/(kg.K) kJ/(kg .K) 2500.9 2500.9 0.0 9.1555 9.1555 9.0783 8.8738 275 280 285 0.001389 0.001001 94.61 49.8 290 0.001920 0.001001 69.63 70.7 295 0.002621 0.001002 51.87 91.7 300 0.003537 0.001003 39.08 112.6 305 0.004719 0.001005 29.77 133.5 310 0.006231 0.001007 22.91 0.001009 17.80 175.3 315 0.008145 320 0.01055 0.001011 13.96 325 0.01 353 0.001013 11.04 330 0.01721 0.001015 8.806 0.02172 0.02719 0.03378 0.04168 0.05108 0.06219 0.07526 0.006231 365 310 325 330 0.001018 7.079 0.001021 5.734 0.001024 4.678 0.001027 3.842 0.001030 3.176 0.001034 2.642 0.001037 2.210 0.001007 22.91 315 0.008145 0.001009 17.80 320 0.01055 0.001011 13.96 0.01 353 0.001013 11.04 0.01721 0.001015 8.806 335 0.02172 0.001018 7.079 0.001021 5.734 340 0.02719 345 0.03378 350 0.04168 355 0.05108 360 0.06219 365 0.07526 0.001024 4.678 0.001027 3.842 0.001030 3.176 0.001034 2.642 370 0.09054 375 0.1083 380 0.1289 385 0.1525 390 0.1796 395 0.2106 400 0.2458 405 0.2856 2496.5 2504.3 0.0283 2484.7 2513.5 0.1041 2472.8 2522.6 0.1784 2461.0 2531.7 0.2513 2449.2 2540.8 0.3228 2437.3 2549.9 0.3931 2425.4 2558.9 0.4622 154.4 2413.5 2567.9 0.5302 2401.6 2576.8 0.5970 196.2 2389.6 2585.7 0.6629 217.1 2377.5 2594.6 0.7277 238.0 2365.4 2603.3 0.7915 258.9 2353.2 2612.1 0.8544 279.8 2340.9 2620.7 0.9164 2328.5 2629.3 0.9775 300.8 321.7 2316.0 2637.7 1.0378 2303.4 2646.1 1.0973 342.7 363.7 2290.7 2654.4 1.1560 2277.8 2662.5 1.2140 384.8 154.4 2413.5 2567.9 0.5302 175.3 2401.6 2576.8 0.5970 196.2 2389.6 2585.7 0.6629 217.1 2377.5 2594.6 0.7277 238.0 2365.4 2603.3 0.7915 258.9 2353.2 2612.1 0.8544 2340.9 2620.7 0.9164 279.8 300.8 2328.5 2629.3 0.9775 321.7 342.7 2303.4 2646.1 1.0973 363.7 2290.7 2654.4 1.1560 2316.0 2637.7 1.0378 384.8 2277.8 2662.5 1.2140 405.8 2264.8 2670.6 1.2713 0.001037 2.210 0.001041 1.859 0.001045 1.572 426.9 2251.6 2678.5 1.3279 0.001049 1.336 0.001053 1.141 469.2 0.001058 0.9794 490.4 2210.9 2701.3 1.4937 0.001062 0.8440 511.7 2197.0 2708.6 1.5477 0.001067 0.7303 532.9 2182.8 2715.7 1.6012 0.001072 0.6345 554.3 2168.3 2722.6 1.6542 448.0 2238.2 2686.3 1.3838 2224.7 2693.9 1.4390 8.6766 8.4862 8.3023 8.1244 7.9523 7.7856 7.6241 7.4674 7.3154 7.1678 7.0244 6.8849 6.7492 6.6171 6.4884 6.3629 6.2405 7.7856 7.6241 7.4674 7.3154 7.1678 7.0244 6.8849 6.7492 6.6171 6.4884 6.3629 6.2405 6.1209 6.0042 5.8900 5.7783 5.6690 5.5619 5.4569 5.3539 9.1066 8.9779 8.8550 8.7375 8.6251 8.5175 8.4145 8.3158 8.2211 8.1303 8.0431 7.9593 7.8787 7.8013 7.7267 7.6549 7.5857 7.5189 7.4545 8.3158 8.2211 8.1303 8.0431 7.9593 7.8787 7.8013 7.7267 7.6549 7.5857 7.5189 7.4545 7.3922 7.3320 7.2738 7.2174 7.1627 7.1096 7.0581 7.0080
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
From the table we can see that the closest temperature to 180C is 1753C The table shows data at inte...View the full answer

Answered By

Brian Kiprono
Taking part in public speeches, blogging, writing essays
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
Calculate the values of S and DG for each of the following processes at 298 K: H2O(l,298 K) H2O(g, V = 1000. L/mol) H2O(l,298 K) H2O(g, V = 100. L/mol) The standard enthalpy of vaporization for...
-
A 3 ft deep fish pond in the shape of a hemisphere has a volume of 56.549 ft3 when it is full. These data have been collected relating depth to volume in the pond: a. Find the equation of the least...
-
Consider the following hypothesis test: H: 45 H: < 45 A sample of 36 is used. Identify the p-value and state your conclusion for each of the following sample results. Use =.01. a. x = 44 and s...
-
Remove left recursion in the following grammar. Show each step. Hint: First remove direct left recursion. Then the indirect left recursion. A AB | Aab | BA| a Bb | | b
-
Distinguish between accounting and financial reporting for state and local governments and VHWOs for the following issues: 1. Measurement focus and basis of accounting 2. Revenue recognition 3....
-
This chapter discussed natural and randomized experiments. How does a natural experiment differ from a randomized one? Which one is likely to yield more accurate results?
-
Let $\mathbf{x}_{j}$ be the $j$ th row of $\mathbf{X}$, and $\mathbf{X}_{-j}$ be the $\mathbf{X}$ matrix with the $j$ th row removed. Show that \[\operatorname{Var}\left[\hat{\beta}_{j}...
-
A pressure-measuring device has been analyzed and can be described by a model with the structure shown in figure a. In other words1 the device responds to pressure changes as if it were a first-order...
-
Whirly Corporation's contribution format income statement for the most recent month is shown below: Sales (8,000 units) Variable expenses Contribution margin Fixed expenses Total Per Unit $ 272,000 $...
-
You are trying to estimate the free cash flow to the firm for RevTech, a technology firm. The firm reported $80 million in earnings before interest and taxes, capital expenditures of $30 million, and...
-
Imagine turning the system of Exercise 2.13 vertically. Repeat the analysis, which now will involve the height of the piston. Using the same values as in Exercise 2.13(b), with D = 8 cm, plot the...
-
How much energy transfer as heat (J) is required to make fluid in a constant volume container undergo a change U = 200 kJ/kg?
-
In 1886 Eugene Goldstein observed positively charged particles moving in the opposite direction to electrons in a cathode ray tube (illustrated below). From their mass, he concluded that these...
-
Figure 1 illustrates how a barrier option works. The stock price starts at $100, ends at $80, and hits the barrier of $75 about halfway through the year. If the option were a 95-strike down-and-in...
-
Consider a directed graph described by the following weighted matrix: W= a. Draw this graph. b. Find the distance matrix using Floyd's algorithm. -0 3 00 - 8 0 00 00 00 8 Loo 00 80 1 8 m 8 3 1 8 1...
-
6. Consider a two-layer neural network as shown in Fig. Q6 with ReLU activation functions in the hidden layer and the output layer. The initial weights, biases and training inputs/outputs for a...
-
The plan is to search the whole record of all rainfall data for a particular station's data, and then present this data on the client screen. This search should be done in a thread. Consider the...
-
Create a Dependency Diagram for the database. For each Determinant give the business rule(s) that created the dependency.
-
Prepare a table like Figure 13-12 using the following information: The forecast for each week of an eight-week schedule is 50 units. The master production scheduling rule is to schedule production if...
-
What is a manufacturing system?
-
Grade 1020 steel has a yield strength of 42 ksi and an elastic modulus of 30 Mpsi. Another grade of steel has a yield strength of 132 ksi. What is its elastic modulus?
-
A steel cable of diameter 3/16 in. is attached to an eyebolt and tensioned to 500 lb (Figure P5.4, see on page 226). Calculate the stress in the cable, and express it in the dimensions psi, ksi, Pa,...
-
When a 120-lb woman stands on a snow-covered trail, she sinks slightly into the snow because the compressive stress between her ski boots and the snow is larger than the snow can support without...
-
Zheng invested $152,000 and Murray invested $252,000 in a partnership. They agreed to share income and loss by allowing a $73,000 per year salary allowance to Zheng and a $53,000 per year salary...
-
On July 31, 2013, Cooks Castle Resorts issues a $2,500,000, 10 year, 6.5% Bond with interest payable semiannually on January 31 and July 31 of each of the next ten years. The market interest rate on...
-
The following transactions are for Sandhill Company, 1 On December 3, Sandhill Company sold 5647,000 of merchandise to Monty Co., on account, terms 2/10, n/30, FOB destination. Sandhill pald $400 for...

Study smarter with the SolutionInn App


