In 1886 Eugene Goldstein observed positively charged particles moving in the opposite direction to electrons in a
Question:
In 1886 Eugene Goldstein observed positively charged particles moving in the opposite direction to electrons in a cathode ray tube (illustrated below). From their mass, he concluded that these particles were formed from residual gas in the tube. For example, if the cathode ray tube contained helium, the canal rays consisted of He+ ions. Describe a process that could lead to these ions. 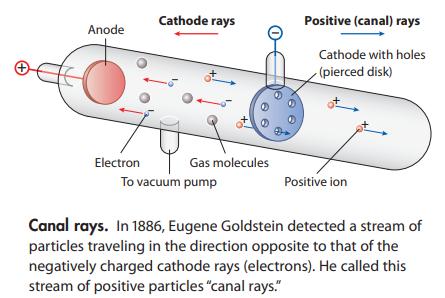
Transcribed Image Text:
Anode Cathode rays Electron To vacuum pump Gas molecules Positive (canal) rays Cathode with holes (pierced disk) Positive ion Canal rays. In 1886, Eugene Goldstein detected a stream of particles traveling in the direction opposite to that of the negatively charged cathode rays (electrons). He called this stream of positive particles "canal rays."
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The process of forming He ions in a cathode ray tube is as follows A high voltage is applied between the cathode and anode creating a strong electric ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Electrons in a cathode ray tube start from rest and are accelerated through a potential difference of 12.0 kV. They are moving in the + x -direction when they enter the space between the plates of a...
-
In a cathode ray tube in an old TV, electrons are accelerated from rest with a constant acceleration of magnitude 7.03 1013 m/s2 during the first 2.0 cm of the tube's length; then they move at...
-
A railroad train is traveling at 30.0 m/s in still air. The frequency of the note emitted by the train whistle is 262 Hz. What frequency is heard by a passenger on a train moving in the opposite...
-
The atoms that constitute your body are mostly empty space, and structures such as the chair you're sitting on are composed of atoms that are also mostly empty space. So why don't you fall through...
-
To create user applications as described in this chapter, systems analysts often use macros. Microsoft defines a macro as a series of commands and instructions that you group together as a single...
-
Todd, who is subject to a 35 percent marginal gift tax rate, made a gift of a sculpture to Becky, valuing the property at $7,000. The IRS later valued the gift at $15,000. Compute the applicable...
-
With reference to Exercise 10.57, find a large sample 95% confidence interval for the true difference of the probabilities of failure. Data From Exercise 10.57 10.57 Two bonding agents, A and B, are...
-
Indicate whether the following statements are true or false. If the statement is false, explain why. a. If a firm repurchases its stock in the open market, the shareholders who tender the stock are...
-
A 200 mm reinforced concrete wall carries a dead load of 140 kN/m including self-weight and a live load of 60 kN/m. It is to be carried by 500 mm thick footing with its bottom 1.5 m below NGL. Soil...
-
Marie Curie was born in Poland but studied and carried out her research in Paris. In 1903, she shared the Nobel Prize in Physics with H. Becquerel and her husband Pierre for their discovery of...
-
Give the complete symbol including atomic number and mass number, of (a). A nickel atom with 31 neutrons, (b). A plutonium atom with 150 neutrons, and (c). A tungsten atom with 110 neutrons. (x7)
-
Find an equation of the line tangent to the following curves at the given point. y 4x x + 3 x = 3
-
Hiro Corp. common stock is selling for $29.50 per share. The last dividend was $2.40 and dividends are expected to grow at an 8% annual rate. Flotation costs on new stock sales are 11% of the selling...
-
The movement of a share price over the next two months is to be modelled using a two-period recombining binomial model. Over each month, it is assumed that the share price will either increase or...
-
Submit the completed Marketing Math Calculations worksheet.Also, please submit a separate Word document in which you answer the following questions: If the retail price is set at $1.00, what effect...
-
Suppose you borrowed $15,000 at a rate of 8% and must repay it in 3 equal installments at the end of each year of the next 4 years. Construct a loan amortization table showing how the loan payments...
-
Your cousin knows you studied finance courses at university, and asks you for information and advice relating to his small mini-mart business. Based on your studies, identify any two (2) finance...
-
Novell, which had a market value of equity of $2 billion and a beta of 1.50, announced that it was acquiring WordPerfect, which had a market value of equity of $1 billion and a beta of 1.30. Neither...
-
What services are provided by the provincial and territorial governments?
-
A refrigerator is operated by a 0.25-hp (1 hp = 746 watts) motor. If the interior is to be maintained at 4.50C and the room temperature is 38C, what is the maximum heat leak (in watts) that can be...
-
Predict the major product obtained upon radical bromination of each of the following compounds: (a) (b) (c)
-
From the following data, calculate ÎH o R,391.4 K for the reaction CH 3 COOH(g) + 2O 2 (g) 2H 2 O(g) + 2CO 2 (g): Values for ÎH o R for the first two reactions are at 298.15 K, and for...
-
An overhead absorption rate of $ 1 2 per direct labour hour was established based on a budget of 2 , 1 0 0 hours. Actual direct labour hours worked were 2 , 1 8 0 and actual overhead expenditure was...
-
Womble, Inc. has beginning inventory of $ 2 0 0 and an ending inventory of $ 4 0 0 for a given period in which it purchased $ 1 3 , 4 0 0 of materials. What is the dollar amount of materials used in...
-
At 1 2 / 3 1 / 2 2 , the end of Jenner Company's first year of business, inventory was $ 6 , 1 0 0 and $ 5 , 1 0 0 at cost and at market, respectively. Following is data relative to the 1 2 / 3 1 / 2...

Study smarter with the SolutionInn App


