Compare the COP of the ammonia refrigeration system of Figure 7.21 to that of the single-stage system
Question:
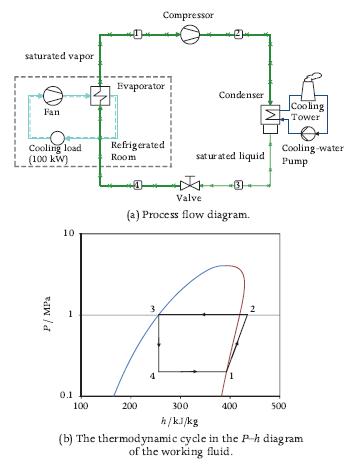
Compare the COP of the ammonia refrigeration system of Figure 7.21 to that of the single-stage system of Figure 7.20, operating with the same working fluid between the same minimum and maximum cycle pressures, and given that the temperature at the outlet of the direct contact heat exchanger equals 40?C. Assume that the isentropic efficiency of all the compressors is the same and is 0.8.
Transcribed Image Text:
saturated vapor Fan Cooling load (100 kW) P/ MPa 10 - 0.1 100 Compressor Evaporator Refrigerated Room 200 Condenser saturated liquid Valve (a) Process flow diagram. 300 h/kJ/kg 3 400 2 Cooling Tower Cooling water Pump 500 (b) The thermodynamic cycle in the P-h diagram of the working fluid.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Properties of NH3 are calculated with STANMIX The notation is that of Figure 720 and 721 Design Data ...View the full answer

Answered By

Parvesh Kumar
I am an experienced Mathematics and Statistics tutor with 10 years of experience teaching students and working professionals. I love teaching students who are passionate to learn subjects or wants to understand any mathematics and statistics concept at graduation or master’s level. I have worked with thousands of students in my teaching career. I have helped students deal with difficult topics and subjects like Calculus, Algebra, Discrete Mathematics, Complex analysis, Graph theory, Hypothesis testing, Probability, Statistical Inference and more. After learning from me, students have found Mathematics and Statistics not dull but a fun subject. I can handle almost all curriculum of mathematics. I did B.Sc (mathematics), M.Sc (mathematics), M.Tech (IT) and am also Gate (CS) qualified. I have worked in various college and school and also provided online tutoring to American and Canadian students. I look forward to discussing with you and make learning a meaningful and purposeful
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
The vortex tube (also known as a Ranque or Hirsch tube) is a device that produces a refrigeration effect by expanding pressurized gas such as air in a tube (instead of a turbine as in the reversed...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Calculate the value of each expression below. Then compare your answers to those in problem 5-90. What do you notice? Homework Help -6+/6-(4)(1)(-40) -6-/6?-(4)(1)(-40) b. a. 2 -1 2-1 -13-/13...
-
A small piece of zinc is dissolved in 50.00 mL of 1.035M HCl. At the conclusion of the reaction, the concentration of the 50.00 mL sample is redetermined and found to be 0.812 M HCl. What must have...
-
Table 6.1 shows how the main categories of federal outlays have changed from 1940 to 2013 (projected). Where have the biggest changes overtime occurred? Where are the biggest changes from 2009 to...
-
Wilderness Products, Inc., has designed a self-inflating sleeping pad for use by backpackers and campers. The following information is available about the new product: a. An investment of $1,350,000...
-
Determine the eigenvalues and eigenvectors of the following equations: a. \(\dot{x}=x-y, \quad \dot{y}=x+3 y\) b. \(\dot{x}=x+y, \quad \dot{y}=4 x+y\)
-
Compute the weighted-average number of shares outstanding for Steadman Company, which has a simple capital structure, assuming that the following transactions in common stock occurred during the...
-
Jeremy (unmarried) earned $100,500 in salary and $6,050 in interest income during the year. Jeremy's employer withheld $10,000 of federal income taxes from Jeremy's paychecks during the year. Jeremy...
-
A 2 -year bond with a semiannual coupon rate of \(4 \%\) per annum is trading at par \((100 \%)\). (a) What is its spot semiannual yield? (b) Assume one can borrow at \(3 \%\) p.a. simple interest...
-
Consider a closed recuperative Brayton cycle turbine power plant employing supercritical CO 2? as the working fluid. The working fluid at the compressor inlet is at T = 32?C and P = 7.7 MPa. CO 2...
-
In the regenerative gas turbine power plant whose process flow diagram is shown in Figure 7.27a, the mass flow of air entering the compressor at 1 bar and 25?C is 17.9 kg/s. The pressure ratio of the...
-
Use the power method to find the largest singular value of the following matrices: (a) (b) (c) (d) 3 3 2 2 2 1 -1 1 -2 0 1 1 121 312
-
State the date (or dates) on which a "large" company would be required to settle its corporation tax liability for each of the following accounting periods: (a) the eleven months to 31 May 2018 (b)...
-
A company's taxable total profits for an accounting period are 3,200,000. Compute the corporation tax liability if the accounting period is: (a) the year to 31 March 2017 (b) the year to 31 March...
-
A company's accounts for the 17 months to 30 June 2017 include: The debentures were acquired (not f or trade purposes) on 1 May 2016 . Interest is payable half -yearly on 30 April and 31 October....
-
Timberlake Ltd prepares accounts to 31 March each year. The company made the following disposals of chargeable assets in the year to 31 March 2018: (i) In February 2018 , a rare Bentley motor car...
-
In 2010, Shaun bought 20% of the ordinary shares of an unlisted trading company. The shares cost 140,000. He owned the shares until January 2018 when he gave all the shares to a friend. On the date...
-
An experimenter places a piece of a solid metal weighing 255 g into a graduated cylinder, which she then fills with liquid mercury. After weighing the cylinder and its contents, she removes the solid...
-
Havel says the grocer doesnt believe what is on the sign and indeed, he says the grocers customers will barely notice it. But Havel maintains that the sign serves a specific function. How would you...
-
Iminodiacetic acid forms 2:1 complexes with many metal ions: A 25.0 mL solution containing 0.120 M iminodiacetic acid buffered to pH 7.00 was titrated with 25.0 mL of 0.050 0 M Cu 2+ .Given that x2...
-
What is the chelate effect?
-
Auxiliary complexing agent. Use the equation derived in Problem 11-19. (a) Prepare a spreadsheet to reproduce the 20-, 50-, and 60-mL points in the EDTA titration of Zn 2+ in the presence of NH 3 in...
-
Years ago, the difference between accounting in commercial (for-profit) organizations and governmental accounting was somewhat simplistically described to me as follows: The goal of accounting in...
-
Sheridan Company leases equipment from Cheyenne Inc. for five years starting on January 1, 2025. The lease is properly classified as a finance/sales-type lease. The terms of the lease are as follows:...
-
9/4-You decided to purchase an office building for your business. The cost of the office was $200,000. Pig E. Bank agreed to finance the purchase and offered you a 30-year 6.0% mortgage and required...

Study smarter with the SolutionInn App


