Obtain charts like those of Figure 6.23 in order to compare values for propane calculated with STANMIX
Question:
Obtain charts like those of Figure 6.23 in order to compare values for propane calculated with STANMIX and TPSI. What can you infer from these charts about the accuracy of the cubic equation of state model if compared to a multi-parameter thermodynamic model?
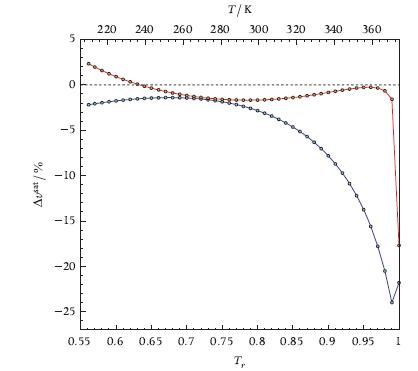
Transcribed Image Text:
Ausat 1% 5 0 -5 | -10 -15 -20 -25 T/K 220 240 260 280 300 0.55 0.6 320 340 360 0.65 0.7 0.75 0.8 0.85 0.9 0.95 Ty 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
The percentage difference between the saturation pressure calculated with Stan...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
Galaxies tend to be strong emitters of Lyman- photons (from the n = 2 to n = 1 transition in atomic hydrogen). But the intergalactic medium the very thin gas between the galaxies tends to absorb...
-
For the data in the table below: i. What do the correlation coefficients of 1, shaded in yellow, indicate? ii. The highest correlation coefficient is 0.84, shaded in green. What can you infer from...
-
You read in a newspaper that the nominal interest rate is 12 percent per year in Canada and 8 percent per year in the United States. Suppose that the real interest rates are equalized in the two...
-
Suppose you needed a material that could absorb heat without having its temperature increase very much. Would you choose aluminum or water? Make sure your explanation uses the concept of specific...
-
Consider an income guarantee program with an income guarantee of $3,000 and benefit reduction rate of 50%. A person can work up to 2,000 hours per year at $6 per hour. Alice, Bob, Calvin, and Deborah...
-
At what temperature will the diffusion coefficient for the diffusion of copper in nickel have a value of 6.5 ? 10 -17 m 2 /s. Use the diffusion data in Table 5.2. Diffusing Species Host Activation...
-
\(\lambda_{1}\) and \(\lambda_{2}\) with same sign \(\left(\lambda_{1}, \lambda_{2} ight.\) : real and distinct) a. Unstable node b. Saddle point c. Node d. Focus or spiral point e. Stable node
-
Douglas C. Mather, founder, chair, and chief executive of Fly-by-Night International Group (FBN), lived the fast-paced, risk-seeking life that he tried to inject into his company. Flying the companys...
-
Your company is looking at a new project in Mexico. The projectwill cost 1,000,000 pesos. The cash flows are expected to be350,000 pesos per year for 5 years. The current spot exchange rateis 19.07...
-
Pavement Corp. ("Pavernent) nas been operating sinoe 1398. Ht is now Docember 31, 2004, and Pavement is about to prepare consolidated finendial etetements for Favement and ts subeidiary. Sigur. Co...
-
Estimate the limiting metastable state for the liquid and the vapor of water at 100C using the van der Waals equation of state.
-
Evaluate the specific volume of heptane for T r = 1.1 and P r = 3.5 using(a) The van der Waals equation of state (T c = 540.13 K, P c? = 27.36 bar);(b) The generalized compressibility chart of Figure...
-
Consider a proportional feedback control system. As shown in Figure 10.47, the closed-loop system becomes marginally stable when the proportional gain is 0.75. Find the P, PI, and PID controller...
-
How do you access the array element with the value of "z"? A. dimensions["three"][2] B. dimensions["three"][3] C. dimensions[2][2] D. dimensions[3][3] dimensions "one" "p" "two" "x" "y" "three" "x"...
-
Given the following application, which specific type of exception will be printed in the stack trace at runtime? A. ClassCastException B. IllegalArgumentException C. NullPointerException D....
-
Which of the following exception classes must be handled or declared in the method in which they are thrown? (Choose three.) A. Happy B. Dopey C. Sleepy D. Sneezy E. Doc F. Grumpy public class Happy...
-
What happens when calling the following method with a non-null and non-empty array? A. It adds an element to the array the value of which is Times Square. B. It replaces the last element in the array...
-
Which fills in the blank in the method signature to allow this code to compile? A. ? extends Collection B. ? implements Collection C. T extends Collection D. T implements Collection E. None of the...
-
A room measures 10.0 ft 11.0 ft and is 9.0 ft high. What is its volume in liters?
-
How does the organizational structure of an MNC influence its strategy implementation?
-
Difference plot. A solution containing 3.96 mmol acetic acid plus 0.484 mmol HCI in 200 mL, of 0.10 M KC1 was titrated with 0.490 5 M NaOH to measure K. for acetic acid. (a) Write expressions for the...
-
Difference plot. A solution containing 3.96 mmol acetic acid plus 0.484 mmol HCI in 200 mL, of 0.10 M KC1 was titrated with 0.490 5 M NaOH to measure K. for acetic acid. (a) Write expressions for the...
-
Why does the solubility of a salt of a basic anion increase with decreasing pH? Write chemical reactions for the minerals galena (PbS) and cerussite (PbCO 3 ) to explain how acid rain mobilizes...
-
1. A high-performing EV is equipped with a lithium battery pack and a bank of supercapacitors. During acceleration, both the battery pack and the supercapacitor provide the required power. The total...
-
Compute the key growth rates of Royal Caribbean and Carnival Cruises. Adjust for the effect of the acquisitions ( Royal acquiring Celebrity and Carnival acquiring Cunard ) on their growth rate in...
-
Suppose that you borrow $17,000 for fourfour years at 6% toward the purchase of a car. Use PMT equals StartStartFraction Upper P left parenthesis StartFraction r Over n EndFraction right parenthesis...

Study smarter with the SolutionInn App


