Obtain the Maxwell relations (6.38c) and (6.38d) starting from the Gibbs equation. Data From ?(6.38c) and (6.38d)
Question:
Obtain the Maxwell relations (6.38c) and (6.38d) starting from the Gibbs equation.
Data From ?(6.38c) and (6.38d)
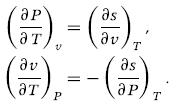
These are known as the Maxwell relations and are valid for simple compressible substances.
Transcribed Image Text:
т T (³7)¸ = (³) ₁. (³7), - - (³²), T P
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
The differential of the Helmholtz energy a u Ts is substitution of ...View the full answer

Answered By

RITU RATHI
I am Ritu Rathi. I have done Master of science (Chemistry). I have always had a passion for tutoring. I believe that Tutors should be concerned about finding the most suitable method of delivering information to students, since each one has a different learning style, so it is important to create an individualized study plan for each student that best fits their method of learning and allows us to make the most of the tutoring experience. I like to teach Organic Chemistry as it is my favorite subject. My aim is to teach Organic Chemistry in simple and easy steps. I put a great effort into explaining concepts until they are well understood. I helped many students in their assignments. My teaching methodology is basically dependent upon breaking down a concept into several smaller ideas so that one can understand it in a better way. I always try to enhance student’s morale and attitude, giving them hope of improvement over the course of tutoring with me.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
The Board of Governors of the Federal Reserve publishes the 1-quarter-ahead forecasts of real GDP growth. These are known as the Greenbook forecasts. Download the realized values of GDP growth (from...
-
Emerson Electric Company was founded in 1890 as a manufacturer of motors and fans. In 1993, Emerson marked its thirty-sixth consecutive year of improved earnings per share. On $8.2 billion sales, the...
-
The purpose of this exercise is to give you an opportunity to create the tables for Bearcat Incorporated. The tables themselves are based on the relations that appear in the following figure. L1:...
-
Give the decoding key for the encoding keys in Problems 2536. Minus 4, divide by 2.
-
Consider the four basic questions of public finance listed in the chapter. Which of these questions are positivequestions that can be proved or disprovedand which are normativequestions of opinion?...
-
The activity of a radioactive sample (with a single radioactive nuclide) decreases to one eighth its initial value in a time interval of 96 days. What is the half-life of the radioactive nuclide...
-
A manufacturer of submersible pumps claims that at most \(30 \%\) of the pumps require repairs within the first 5 years of operation. If a random sample of 120 of these pumps includes 47 which...
-
Presented below and on page 348 are the purchases and cash payments journals for Richmond Co. for its first month of operations. In addition, the following transactions have not been journalized for...
-
The total payroll of Crane Company for October, 2025 was $788000, of whic $147000 represented amounts paid in excess of $128,400 to certain employees for FICA. $490000 represented amounts paid to...
-
You have a 9 9 grid of squares, each of which can be colored red or blue. The grid is initially colored all blue, but you can change the color of any square any number of times. Imagining the grid...
-
Given the truncated virial equation for calculate the pressure for T = 180 K and v = 0.03 m 3 /kg and calculate the specific volume for P = 0.3 MPa and T = 320 K. Compare the values with the values...
-
Determine the reference state for the entropy and enthalpy of R134a chosen for the model implemented in STANMIX by inspecting the thermodynamic T ?? s or P ? h diagram in Appendix A. Determine the...
-
As noted on page 283, when the two population means are equal, the estimated standard error for the independent-measures t test provides a measure of how much difference to expect between two sample...
-
You are given the following information with respect to a bond: ( i ) face value: 1 0 0 ( ii ) term to maturity: 1 . 5 years ( iii ) annual coupon rate: 5 % payable semiannually You are also given:...
-
If the economy booms, R&F, Inc. stock is expected to return 16%. If the economy goes into a recessionary period, then R&F is expected to only return 3%. The probability of a boom is 80% while the...
-
Arrange the following items in proper balance sheet presentation: Note: Amounts to be deducted should be indicated with parentheses or a minus sign. Accumulated depreciation Retained earnings Cash...
-
A company incurs $1210000 of overhead each year in three departments, Processing, Packaging, and Testing. The company performs 1000 processing transactions, 200000 packaging transactions, and 1900...
-
Stellar Sound, Inc., which uses a job-order costing system, had two jobs in process at the start of 20x1: job no. 64 ($84,100) and job no. 65 ($53,500). The following information is available: a. The...
-
How much money was needed 15 years ago to have the purchasing power of $1000 today if the (compound annual) rate of inflation has been: a. 2%? b. 4%?
-
What is master production scheduling and how is it done?
-
(a) A 3.96 10 4 M solution of compound A exhibited an absorbance of 0.624 at 238 nm in a 1.000-cm cuvet; a blank solution containing only solvent had an absorbance of 0.029 at the same wavelength....
-
Ammonia can be determined spectrophotometrically by reaction with phenol in the presence of hypochlorite (OCl - ): 4.37-mg sample of protein was chemically digested to convert its nitrogen into...
-
Cu + reacts with neocuproine to form the colored complex (neocuproine)2Cu + , with an absorption maximum at 454 nm. Neocuproine is particularly useful because it reacts with few other metals. The...
-
A project that costs $4,450 to install will provide annual cash flows of $1,500 for each of the next 6 years. a. What is NPV if the discount rate is 11%? b. How high can the discount rate be before...
-
Prove MM1: Both Umbrella Inc. and Prixxa Inc. produce $300,000 of cash flow each year. Umbrella has no debt outstanding, and its cost of equity capital is 14 percent. But Prixxa has 1,000,000 depts...
-
How did they find the over head rate percentages for current year and last year? A job order cost sheet for Cullumber Company is shown below. Job No. 92 For 2,000 Units Date Direct Materials Direct...

Study smarter with the SolutionInn App


