One kilogram of R134a is heated in a constant volume container from the critical point to 450
Question:
One kilogram of R134a is heated in a constant volume container from the critical point to 450 K. How much energy is transferred as heat to the fluid? Use the diagrams or data in Appendix A.4.
Data From A.4
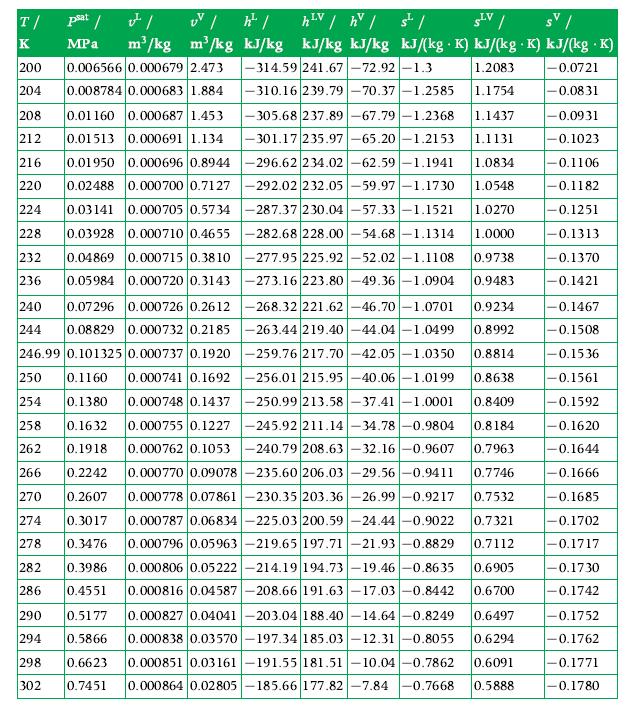
Transcribed Image Text:
T/ K 200 204 h¹V / hv / s¹/ LV 282 286 psat / ¹/ UV /h¹/ SLV / SV/ MPa m³/kg m³/kg kJ/kg kJ/kg kJ/kg kJ/(kg K) kJ/(kg .K) kJ/(kg .K) -314.59 241.67-72.92 -1.3 1.2083 -0.0721 -310.16 239.79-70.37 -1.2585 1.1754 -0.0831 0.006566 0.000679 2.473 0.008784 0.000683 1.884 208 0.01160 0.000687 1.453 -305.68 237.89-67.79-1.2368 1.1437 212 0.01513 -301.17 235.97-65.20 -1.2153 1.1131 216 0.01950 0.000691 1.134 0.000696 0.8944 -296.62 234.02-62.59 -1.1941 1.0834 0.000700 0.7127 -292.02 232.05-59.97 -1.1730 1.0548 220 0.02488 224 0.03141 0.000705 0.5734-287.37 230.04-57.33 -1.1521 1.0270 228 0.03928 232 0.04869 0.05984 236 0.1918 0.000710 0.4655 -282.68 228.00 -54.68 -1.1314 1.0000 0.000715 0.3810 -277.95 225.92-52.02 -1.1108 0.9738 0.000720 0.3143 -273.16 223.80-49.36-1.0904 0.9483 240 0.07296 0.000726 0.2612 -268.32 221.62 -46.70-1.0701 0.9234 244 0.08829 0.000732 0.2185 -263.44 219.40-44.04 -1.0499 0.8992 246.99 0.101325 0.000737 0.1920 -259.76 217.70-42.05 -1.0350 0.8814 250 0.1160 0.000741 0.1692 -256.01 215.95 -40.06-1.0199 0.8638 254 0.1380 0.000748 0.1437 -250.99 213.58 -37.41 -1.0001 0.8409 258 0.1632 0.000755 0.1227 -245.92 211.14-34.78 -0.9804 0.8184 262 0.000762 0.1053 -240.79 208.63 -32.16 -0.9607 0.7963 266 0.2242 0.000770 0.09078 -235.60 206.03-29.56 -0.9411 0.7746 270 0.000778 0.07861-230.35 203.36 -26.99 -0.9217 0.7532 274 0.3017 0.000787 0.06834 -225.03 200.59-24.44 -0.9022 0.7321 278 0.3476 0.000796 0.05963 -219.65 197.71-21.93 -0.8829 0.7112 0.000806 0.05222 -214.19 194.73-19.46 -0.8635 0.6905 0.000816 0.04587 -208.66 191.63-17.03 -0.8442 0.6700 290 0.5177 0.000827 0.04041 -203.04 188.40-14.64 -0.8249 0.6497 294 0.5866 0.000838 0.03570-197.34 185.03-12.31 -0.8055 0.6294 298 0.6623 0.000851 0.03161-191.55 181.51 -10.04 -0.7862 0.6091 302 0.7451 0.000864 0.02805 185.66 177.82 -7.84 -0.7668 0.5888 0.2607 0.3986 0.4551 -0.0931 -0.1023 -0.1106 -0.1182 -0.1251 -0.1313 -0.1370 -0.1421 -0.1467 -0.1508 -0.1536 -0.1561 -0.1592 -0.1620 -0.1644 -0.1666 -0.1685 -0.1702 -0.1717 -0.1730 -0.1742 -0.1752 -0.1762 -0.1771 0.1780
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
1 Make an idealized sketch of the system and identify the boundary indicating if it moves in time 2 If the boundary moves indicate the forces acting o...View the full answer

Answered By

Muhammad Zain
I did matric in 2017 from high school. I also did inter in 2019 from govt college. I get admission in university for graduation. Now I am student that time . I have two years experience for teaching at academy.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
In R2, where find the change of basis matrices from D to ε2 and from ε2 to D. Multiply the two. 4
-
A hospital wants to hire 3 doctors in 3 different specialties. There are 10 applicants. How many choices are there in each of the following cases: a). All candidates have all 3 specialties. b). One...
-
A gas mixture of 1 kmol carbon monoxide, 1 k mol nitrogen, and 1 k mol oxygen at 25C, 150 kPa, is heated in a constant pressure SSSF process. The exit mixture can be assumed to be in chemical...
-
When are objects on the periphery of your vision most noticeable?
-
Your portfolio has a beta equal to 1.3. It returned 12% last year. The market returned 10%; the risk-free rate is 6%. Calculate Treynors measure for your portfolio and the market. Did you earn a...
-
The cycle in Figure represents the operation of a gasoline internal combustion engine. Volume V3 = 4.00V1. Assume the gasoline - air intake mixture is an ideal gas with = 1.30 what are the ratios...
-
Outline the project planning process based on identified resources.
-
McCormick Corporation issued a 4-year, $40,000, 5% note to Greenbush Company on January 1, 2011, and received a computer that normally sells for $31,495. The note requires annual interest payments...
-
A 305 N unbalanced force was applied to an 25.0 kg object at rest. How far would this object travel after 8.00 seconds? Remember to identify all your data, write the equation, and show your work.
-
One kilogram of carbon dioxide is expanded adiabatically in a pistoncylinder system from 10 bar and 500 K to the saturated-vapor state at 5 bar. How much work is done by carbon dioxide and on what?...
-
(a) Graph f(x) = e x 2 and shade the area represented by the improper integral e x 2 dx. (b) Use a calculator or computer to find a a e x 2 dx for a = 1, a = 2, a = 3, a = 5. (c) The improper...
-
A well-known method of generating a sequence of "pseudo-random" integers x0, x1. . . . . . . in the interval from 0 to n is based on the Fibonacci equation u(k+2) = u(k+1) + u(k) mod n. with initial...
-
In the first payment on a 60-month, $10,000 car loan with APR of 6.51%, how much pays off the principal? Round your answer to the nearest cent (one-hundredth). Do not include the dollar sign, $.
-
What political issues did the investigators in the Dworkin chapter have to overcome? How would you have addressed them if you were the one investigating the cholera outbreak?
-
Astro Company sold 23,500 units of its only product and reported income of $186,000 for the current year. During a planning session for next year's activities, the production manager notes that...
-
What will be printed when the following statements are executed? x = 5; System.out.print("1 "); if (x == 6) System.out.print("2"); else System.out.print("3"); System.out.print("4");
-
A center pivot sprinkler system costing $997 per acre irrigates 117 acres of alfalfa, which requires an annual net application of 53 inches. What is the total annual cost for labor and maintenance if...
-
Describe how you would separate the metal ions in a solution containing silver ion, copper(II) ion, and nickel(II) ion, using the sulfide scheme of qualitative analysis.
-
Read the Forecasting Supply Chain Demand Starbucks Corporation case in your text Operations and Supply Chain Management on pages 484-485, then address the four questions associated with the...
-
The grand potential (T, V, {A}), also known as the Landau free energy, is a thermodynamical potential obtained by performing Legendre transformations ofthe internal energy U(S, V, {NA}). Use Legendre...
-
Establish the expression of the differential of the internal energy dU S (T, V), V as a function of the temperature T and the volume V. In the particular case of a gas that satisfies the relation pV...
-
A gas is characterised by the enthalpy H(S, p) = C p T, where C p is a constant (called heat capacity and defined in 5.2), and by pV = NR T, where p is its pressure, V its volume, T its temperature...
-
Loquat County uses a fiscal agent to distribute payments of interest to bondholders. Interest payments are due on July 1 of each year. The fiscal agent requires the county to transfer by June 20 the...
-
. An electron accelerates from rest through an electric field and into a magnetic field travelling at a speed of 3 x 10 m/s as shown in the diagram below. The plates have a potential difference of 25...
-
$50,570 payments to be made at the end of each period for 17 periods at 10%. (Round factor values to 5 decimal places, e.g. 1.25124 and final answer to O decimal places, e.g. 458,581.) Present value...

Study smarter with the SolutionInn App


