The cycle in Figure represents the operation of a gasoline internal combustion engine. Volume V3 = 4.00V1.
Question:
The cycle in Figure represents the operation of a gasoline internal combustion engine. Volume V3 = 4.00V1. Assume the gasoline - air intake mixture is an ideal gas with γ = 1.30 what are the ratios
(a) T2/T1
(b) T3/T1,
(c) T4/T1
(d) p3/p1,
(e) p4/p1?
(f) What is the engine efficiency?
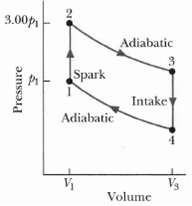
3.00p Adiabatic Spark Intake Adiabatic V3 Volume Pressure
Step by Step Answer:
a The pressure at 2 is p2 300p as given in the problem statement The volume is V VnRTp The temperatu...View the full answer



Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Related Video
An air pump is a device used to inflate or deflate objects by moving air in or out of them. There are many different types of air pumps, ranging from manual hand pumps to electrically powered pumps, and they are used for a variety of purposes. John had trouble blowing up many balloons without a balloon pump and experienced pain. He solved this problem by using an empty plastic bottle with a rectangular-shaped hole in the cap, a cut piece of balloon stretched over it, and a small hole in the bottle. By pressing and releasing the bottle, he created an air pump to inflate the balloons without any discomfort. Air pumps utilize fluid mechanics and thermodynamics principles to increase air pressure inside an object being inflated, such as a bicycle tire. The ideal gas law is used to calculate the pressure inside the tire. As air pressure increases, the object becomes more rigid and can support more weight. The efficiency of the pump is determined by how much of the work done is converted into useful work, such as inflating the tire, and how much is lost as heat. Understanding these principles is important to effectively use and optimize the use of air pumps.
Students also viewed these Thermodynamics questions
-
An ideal gas with the adiabatic exponent goes through a cycle (Fig. 2.3) within which the absolute temperature varies -fold. Find the efficiency of this cycle.
-
An ideal gas with the adiabatic exponent y goes through a process p = Po- aV, where Po and a are positive constants, and V is the volume. At what volume will the gas entropy have the maximum value?
-
An ideal gas with k = 1.4 is flowing through a nozzle such that the Mach number is 2.4 where the flow area is 25 cm2. Assuming the flow to be isentropic, determine the flow area at the location where...
-
Two kilograms of water, initially saturated liquid at 10 kPa, are heated to saturated vapor while the pressure is maintained constant. Determine the work and the heat transfer for the process, each...
-
Why is it that the greater the number of firms is in a monopolistically competitive industry, the lower the price is, but the higher the average cost of each firm is for a given level of output?
-
During an economic slowdown, an automobile plant lost $12,000,000 on the production and sale of 9000 cars. Total revenue for the year was $270,000,000. If the break-even volume for the plant is...
-
List five topics which are usually referred to in audit engagement letters.
-
(Qualitative Characteristics) The qualitative characteristics that make accounting information useful for decision-making purposes are as follows. Identify the appropriate qualitative...
-
(a)How high (in m) a hill can a car coast up (engine disengaged) if friction is negligible and its initial speed is 86.0 km/h? (b)If, in actuality, a 750 kg car with an initial speed of 86.0 km/h is...
-
In his autobiography A Sort of Life, British author Graham Greene described a period of severe mental depression during which he played Russian Roulette. This "game" consists of putting a bullet in...
-
In the first stage of a two-stage Carnot engine, energy is absorbed as heat Q1 at temperature T1, work W1 is done, and energy is expelled as heat Q2 at a lower temperature T2. The second stage...
-
The electric motor of a heat pump transfers energy as heat from the outdoors, which is at 5.0oC, to a room that is at 17oC.If the heat pump were a Carnot heat pump (a Carnot engine working in...
-
Refer to Original Source 9 on the companion website, Suicide Rates in Clinical Trials of SS-RIs, Other Antidepressants, and Placebo: Analysis of FDA Reports. Table 1 in that paper (page 791) provides...
-
(a) why you decided to pursue this programme human resource administration of study, (b) explain what you knew about the subject human resource administration, when you started the semester, (c)...
-
In your response post, discuss how you would have felt if you were the other person in the interaction described. Explain what you feel could happen if a client were to feel the same way. To be...
-
Case - Seaworld of Florida v. Perez Book - Employment Law for Human Resource Practice 6th Edition 1. What are the legal issues in this case? What did the appeals court decide? 2. What things must be...
-
Use PESTEL to identify and critically analyse AUS Amazon's business strategy (cost leadership, differentiation or hybrid) in the following sector: e-commerce. write a Business Report that must...
-
create floor plan/design of a literacy centre at a childcare centre. What might it look like for you? Refer to Chapter 10: developing literacy centers of the Creating Environments for Learning...
-
Let \(y=\) expenditure ( \(\$\) ) on food away from home per household member per month in the past quarter and \(x=\) monthly household income (in hundreds of dollars) during the past year. a. Using...
-
Use Stokes' Theorem to evaluate f(y+sin x) dx+(z+cos y) dy+rdz, where C is the rve r(t) = (sint, cost, sin 2t), t = [0, 2].
-
A small pickup truck that has a camper shell slowly coasts toward a red light with negligible friction. Two dogs in the back of the truck are moving and making various inelastic collisions with each...
-
Modern vacuum pumps make it easy to attain pressures of the order of 10-13 atm in the laboratory. (a) At a pressure of 9.00 X 10-14 atm and an ordinary temperature of 300.0 K, how many molecules are...
-
The Lagoon Nebula (Fig. 18.28) is a cloud of hydrogen gas located 3900 light-years from the earth. The cloud is about 45 light years in diameter and glows because of its high temperature of 7500 K....
-
In a gas at standard conditions, what is the length of the side of a cube that contains a number of molecules equal to the population of the earth (about 6 X 109 people)?
-
Do you envision the business as starting by targeting specific needs of a single target community (e.g. the target is San Francisco), targeting specific needs of a similar set of communities (e.g....
-
Define social media. ( 20 points ) How has social media changed the way marketers and consumers communicate with one another? Explain the implications for marketers. Describe how marketing managers...
-
Find an article or video offering advice on choosing the best financing option for a business. Answer the following questions: Which types of financing did they discuss? What advice did they offer?...

Study smarter with the SolutionInn App


