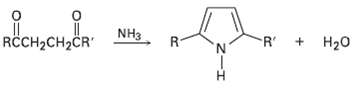
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R +
Question:
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism.
Transcribed Image Text:
R + H20 RCCH2CH2ČR" R- NH3 R.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
HCCH 1 RC CR 11 O HCCH 11 1 RC HA HA 0NH proton N atio...View the full answer

Answered By

Sadoc Raju
I have been teaching High School Physics in India for the last nine years of my life.
I have taught in State Board, CBSE, ICSE and IGCSE schools.
I have gained incredible experience and valuable insight over these years having worked in five different schools in two states with a diverse school population.
I have been certified by Pune University, considered to be the premier institute for education in India.
I also have worked as an aircraft technician to gain practical knowledge in mechanics and aerospace.
Why do I teach?
For me, the rewards and challenges of teaching offer a sense of satisfaction that is enhanced by the knowledge that I have had some impact on the academic and personal development of the children I teach. I believe in developing with the children a supportive classroom environment, which has a basis in quickly developing rapport with the children. I pride myself to be a dedicated self-motivated achiever who is committed to success and adapt at juggling multiple tasks in a high-pressured environment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Ammonia is a principal nitrogen fertilizer. It is prepared by the reaction between hydrogen and nitrogen. In a particular reaction, 6.0 moles of NH3 were produced. How many moles of H2 and how many...
-
Ammonia is compressed from 120 kPa with x = 1 to a pressure of 1.2 MPa and a temperature of 100C. For a mass flux of 3 kg/s, determine the power required to drive the adiabatic compressor.
-
A piston/cylinder contains 1 kg of ammonia at 20C with a volume of 0.1 m3, shown in Fig. P5.129. Initially the piston rests on some stops with the top surface open to the atmosphere, Po, so a...
-
A fast-food chain randomly attaches coupons for prizes to the packages used to serve french fries. Most of the coupons say Play again, but a few are winners. Seventy-five percent of the coupons pay...
-
In what ways does Costco meet the criteria for a "sustainable" organization?
-
Here are mass spectrometric signals for methane in H2: a) Subtract the blank value (9.1) from all other values. Then use the method of least squares to find the slope and intercept and their...
-
The independence of an internal auditing department will most likely be assured if it reports to the: a. President. b. Controller. c. Treasurer. d. Board of directors. Choose the correct answer.
-
Nona Curry started her own consulting firm, Curry Consulting Inc., on May 1, 2017. The following transactions occurred during the month of May. May 1 Stockholders invested $15,000 cash in the...
-
Calculate the magnitude (signs don't matter) of the centripetal acceleration in an electron if it is moving around a nucleus with a speed of 2.18 x 10 m/s. The diameter of the electron's orbit is...
-
Determine the force in members GF, CF, and CD of the roof truss and indicate if the members are in tension or compression. 1.5 kN 1.70 m 2 kN 1.5 m 0,8 m G. -1 m- 2 m 2 m
-
Protonation of an amide using strong acid occurs on oxygen rather than on nitrogen. Suggest a reason for this behavior, taking resonance intoaccount. :0: H2SO4 NH2 NH2 R.
-
3, 5-Dimcthylisoxazole is prepared by reaction of 2, 4-pentanedionc with hydroxylamine. Propose amechanism. CH CH3CCH2CCH3 + H2NOH 3,5-Dimethylisoxazole
-
Evaluate the integral. 1 - tan 0 - de 1 + tan 0
-
The x-intercept of the graph of 9x + 3y = 27 is (Type
-
Simplify. 5 3ns 3 7 2m n 3
-
Incomplete A/R T-account for the month of July is as follows: Dr. Accounts Receivable Cr. 1-July 26000 7-July 140000 31-July 50000 Assuming that all debits to A/R represent sales on account, how much...
-
Rewrite using only positive rational exponents. 6 5 5 7
-
The weights of adult individuals in a certain country are normally distributed with a population mean of =172 pounds and a population standard deviation of = 29 pounds. Suppose n = 36 individuals are...
-
What is the first step in an analysis of financial statements? (a) Check the auditors report. (b) Check references containing financial information. (c) Specify the objectives of the analysis. (d) Do...
-
Establish identity. cos( + k) = (-1)k cos , k any integer
-
Write the formula for the conjugate base of each acid. a. HCI b. HSO3 c. HCHO, d. HF
-
What are all of the structural possibilities for C4H6? (Nine compounds, four acyclic and five cyclic, are known.)
-
Write an equation for each of the following reactions: a. 2-butene + HCl b. 3-hexene + HI c. 4-methylcyclopentene + HBr
-
Use Markovnikov's Rule to predict which regioisomer predominates in each of the following reactions: a. 1-pentene + HBr b. 2-methyl-2-hexene + H2O (H+ catalyst)
-
What logic gate does this represent (reduce it) Do D
-
What basic logic gate does this implement? a b in out Not otb E in Not O out nota And out aAndNotb out Or out b notAndb a out And b
-
Design a Fuzzy system to reduce effect of impulse noise on a noisy image with intensities ranging from [0, L-1]. Let zs denotes the intensity of middle pixel of the neighborhood. The output intensity...

Study smarter with the SolutionInn App


