Protonation of an amide using strong acid occurs on oxygen rather than on nitrogen. Suggest a reason
Question:
Protonation of an amide using strong acid occurs on oxygen rather than on nitrogen. Suggest a reason for this behavior, taking resonance intoaccount.
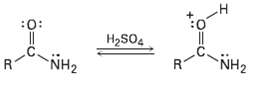
Transcribed Image Text:
:0: H2SO4 NH2 NH2 R.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
0 NH3 NH NProtonation ...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
An equimolar mixture of oxygen and nitrogen enters a compressor operating at steady state at 10 bar, 220 K with a mass flow rate (m) of 1 kg/s. The mixture exits the compressor at 60 bar, 400 K with...
-
A friend of yours has performed three titrations: strong acid with a strong base, weak acid with a strong base, and weak base with a strong acid. He hands you the three titration curves, saying he...
-
Using the curved-arrow notation, derive a resonance structure for the allyl anion (shown here) which shows that the two carbon-carbon bonds an identical bond order of 1.5 and that the unshared...
-
You recently joined an accounting consulting firm. Your first client is a new manufacturing start-up that is trying to set up their costing system, and develop good processes for their upcoming...
-
What would you describe as Costco's basic strategy as a retailer? How do human resource practices support that strategy?
-
Figure 16P-6 shows a local labor market for mechanics. What are the quantity supplied and quantity demanded when the minimum wage is each of the following: a. $40,000? b. $20,000? c. $60,000 Figure...
-
The probability that a wafer contains a large particle of contamination is 0.01. If it is assumed that the wafers are independent, what is the probability that exactly 125 wafers need to be analyzed...
-
A firm is concerned about the condition of some of its plant machinery. Bill James, a newly hired engineer, was assigned the task of reviewing the situation and determining what alternatives are...
-
3. Solve system of linear equations using either Gaussian elimination with back-substitution. Show your work (explain what equations you are multiplying and/or adding when apply). 4x+5y-5z = -23 -2y...
-
Organic Chemistry Structure and Function by Peter Vollhardt and Neil Schore 6th Edition, exercise 19-9. Someone please teach me. Exercise 19-9 Try It Yourself Using chemical equations, show how you...
-
Oxazole is a live-membered aromatic heterocycle. Would you expect oxazole to be more basic or less basic than pyrrole?Explain. Oxazole N:
-
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R + H20 RCCH2CH2R" R- NH3 R.
-
Think of two physical things in your home or office that, if they were embedded with sensors and linked to a network, would improve the quality of your work or personal life. Describe these two...
-
How can leaders foster a culture of collaboration and knowledge sharing within their teams, promoting interdisciplinary perspectives, cross-functional expertise, and a collective commitment to...
-
Evaluate Walmart's globalization strategy over the last two decades. Where did the retailer struggle? Where did it do well? Walmart entered in some countries through acquisition and in some other...
-
Discuss some of the common elements of tables and how you would approach the table design. Discuss the relationship types and how they affect your design. Explain primary key and foriegn key and the...
-
Explain the majorbreakthrough that was achieved by the BSM model Why do we assume alognormaldistribution for stock prices - what is the value of this approach? Explain the concept of implied...
-
explain the concept of put-callparity in simple terms How could a breakdown in the put-call parity relationship provide an arbitrage opportunity in the market? Provide a simple, mathematical example...
-
The financial balances for Steves Car Rentals on 31st May, 2019 are provided below in a table in accounting equation format. You are required to: a draw up the table and list the balances for May b...
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
Write the formula for the conjugate acid of each base. a. NH3 b. CIO C. HSO4 2- d. CO3-
-
As noted in the "A Word about . . . Methane, Marsh Gas, and Miller's Experiment" on page 60, methane can be formed in muddy sediments because of the reducing environment (i.e., lack of oxygen). Write...
-
Explain why 1,3-difluorobutane is a correct IUPAC name, but 1,3-dimethylpentane is not a correct IUPAC name.
-
Draw Newman projections for two different staggered conformations of butane (looking end-on at the bond between carbon-2 and carbon-3), and predict which of the two conformations is more stable. (If...
-
Mizan Corporation, a manufacturing company, prepares its financial statements annually. During the last month of the fiscal year, Mizan experiences a significant surge in sales. The sales team closes...
-
A firm uses job - order costing. Current period facts: Actual overhead: $ 1 0 4 , 6 8 8 Applied overhead: $ 1 0 4 , 9 0 5 Budgeted overhead: $ 1 0 8 , 6 9 8 COGS balance ( before closing ) : $ 3 9 0...
-
On January 1 , 2 0 X 2 P pays $ 1 0 0 million to acquire 5 0 , 0 0 0 of the common voting shares of S , which gives P a 2 5 % ownership interest in S . The January 1 , 2 0 X 2 book value of S ' s...

Study smarter with the SolutionInn App


