Tell how much hydrogen is bonded to each carbon in the following compounds, and give the molecular
Question:
Tell how much hydrogen is bonded to each carbon in the following compounds, and give the molecular formula of each substance:
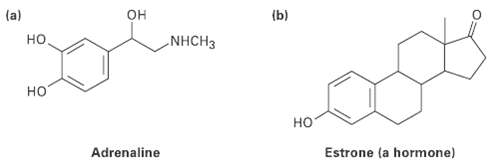
Transcribed Image Text:
(b) он NHCH3 но но но Adrenaline Estrone (a hormone)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Strategy Remember that the end of a line represents a carbon atom with 3 hydrogen...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Hydrogen is bonded to fluorine in hydrogen fluoride by a covalent bond. Write a Lewis formula for hydrogen fluoride.
-
Give the molecular formula of a cyclic alkane, a cyclic alkene, alinear alkyne, and an aromatic hydrocarbon that in each case contains six carbon atoms.Which are saturated and which are unsaturated...
-
Hydrogen is bonded to fluorine in hydrogen fluoride by a covalent bond. Write a Lewis formula for hydrogen fluoride.
-
Why is it likely to have preexisting normal faults in an orogenic belt?
-
During his senior year in college, Sandy is drafted by the Los Angeles Dodgers. When he graduates, he expects to sign a five-year contract in the range of $1.7 million per year. Sandy plans to marry...
-
Shown below is a synthesis of the elm bark beetle pheromone, multistriatin (Problem 16.44). Give structures for compounds A, B, C, and D. TsC OH LiAIH, base Lewis acid C (C10H1BO) Multistriatin
-
Air is flowing in the converging-diverging nozzle shown in Fig. P11.52. Determine the three critical pressure ratios and the Mach numbers immediately upstream and immediately downstream from the...
-
Equation 3.9 indicates that the choice as to whether to convert from a deductible IRA into a Roth IRA when the taxpayer expects his future tax rate to decline depends on the relative magnitude of the...
-
Respond in a summative manner to the 2 separate posts. Post 1 In your reading it sets forth the pronouncement that the Constitution is the supreme law of the land. How does Planned Parenthood of S.E....
-
A non-EU European country has recently been paralyzed by the direct action of truck drivers who are angry at a package of regulations that the government is considering introducing. Discussion...
-
Identify all nonbonding lone pairs of electron in the following molecules, and tell what geometry you expect for each pf the indicated atoms. (a) The oxygen atom in the dimethyl ether, CH3 ? O ? CH3...
-
Propose skeletal structures for compounds that satisfy the following molecular formulas. There is more than one possibility in each case. (a) C5H12 (b) C2H7N (c) C3H6O (d) C4H9Cl
-
Using the Black ScholesMerton formula, find the price of a call option.
-
Notwithstanding that many books are now electronic (or digital) products, in 2020, physical books still accounted for approximately what percent of total book sales?
-
According to lecture, our visual perception is strongly based on our own life experiences. This makes me wonder, how can we become more aware of our own visual perceptions of the world? Further, what...
-
Kayla and David have been married for five years and have no dependent children. They separated on 4 March 2023. They have both been covered by the same private health insurance policy for the fully...
-
Heterogeneous reactions between gas and solid catalysts will be done to produce syngas. There are 3 kinds of reactors used to get syngas, namely Fluidized Bed, Fixed Bed, and Fix Bed Multi Tube....
-
"disruptive technologies" in the last five years. Discuss how the Lyft company was able to gain a competitive advantage. Discuss the company's value proposition and how it successfully gained an...
-
The market-adjusted cost in basis points for the sell order of West Commerce shares is closest to a: A. cost of 249 bps. B. savings of 50 bps. C. savings of 68 bps. Michelle Wong is a portfolio...
-
For the data in Exercise 17-19, use the FIFO method to summarize total costs to account for, and assign these costs to units completed and transferred out, and to units in ending work in process....
-
Compute the partial molar volumes of methyl formate in methanolmethyl formate and ethanolmethyl formate mixtures at 298.15 K for various compositions using the experimental data in Fig. 8.1-2a and...
-
Draw a structure for each model. Explain whether the molecule represented by each model has a dipole moment or not.
-
Draw a structure for each model. Show the direction of the dipole moment for the molecule represented by each model.
-
Draw a structure for the model, N the molecule polar or non-polar explain?
-
ABC Hospital is one of two hospitals in the community. The Hospital offers all major specialty services (e.g. cardiology, neurology, pulmonology, etc.) The Hospital is approached by a cardiologist...
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
On January 8, the end of the first weekly pay period of the year, Regis Company's employees earned $27,760 of office salaries and $70,840 of sales salaries. Withholdings from the employees' salaries...

Study smarter with the SolutionInn App


