Tell whether each of the following reactions is an oxidation, a reduction, orneither: (a) CH3CH CH3CH2OH
Question:
Tell whether each of the following reactions is an oxidation, a reduction, orneither:
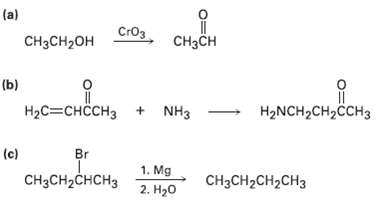
Transcribed Image Text:
(a) Стоз CH3CH CH3CH2OH (b) HаС—снссHз + NHз — НаNCH2CH2CCH3 Br (c) 1. Mg CHзCH2CHCHз CняCH2CH2CH3 2. H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a This reaction is an oxidation b The reac...View the full answer

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Indicate whether each of the following reactions is an oxidation reaction, a reduction reaction, or neither: a. b. c. d. e. f. H2 CHj 3CI partially deactivatedCH Pd HBr RCH CHRRCH2CHR Br2 H2CrO4...
-
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB, orE2: NaN3 (a) CHCH2CH2H2Br CH3CH2CH2CH,N=N=N THE CI (b) , CCH-CHCH2CH CHCH2CHCHCH3 Ethanol (c) H CI -CH -- -CH (d)...
-
Tell wbether each of the following reactions favors reactants or products at equilibrium. (Assume that all reactants and products are soluble.) (a) CH3CI + I- CH3I + CI- (b) CH3CI + -OCH3 CH3OCH3 +...
-
Scranton Refrigeration Corporation began operations at the beginning of the current year. One of the companys products, a compressor, sells for $370 per unit. Information related to the current years...
-
Refer to the BMW Company case. Design a spreadsheet that will allow the firm's managers to estimate what percentage of the firm's net income 10 years into the future will be devoted to disposal of...
-
How did Rutherford interpret the following observations made during his -particle scattering experiments? (a) Most particles were not appreciably deflected as they passed through the gold foil. (b)...
-
1. What are some ways that eBay is trying to improve its performance is by using AI to create new and helpful applications designed to make shopping more fun? 2. Why is website search so important...
-
Based in Italy, Datura, Ltd., is an international importer-exporter of pottery with distribution centers in the United States, Europe, and Australia. The company was very successful in its early...
-
XYZ Inc is a Canadian Controlled private corporation. The company's controller has prepared an income statement for the year for the company(the amounts shown do not include any applicable HST): XYZ...
-
The following financial statements were prepared on December 31, Year 6. Additional Information: Pearl purchased 80% of the outstanding voting shares of Silver for $3,300,000 on July 1, Year 2, at...
-
Which of the following compounds have the same oxidation level, and which have differentlevels? OH 5.
-
How would you carry out the followingsyntheses? Cyclohexene Cyclohexanol Cyclohexane ~/~/al
-
Show that the surface area of a sphere of radius a is still 4a 2 by using Equation (3) to find the area of the surface generated by revolving the curve y = a 2 - x 2 , -a x a, about the x-axis. dy...
-
4. Assume that the number of COVID-19 patients increases according to the law of exponential growth. There were 110 patients after the third day of the pandemic and 460 patients after the seventh...
-
Traditionalist ( 7 6 to 9 9 years old ) . Baby Boomer ( 5 7 to 7 5 years old ) . Generation X ( 4 1 to 5 6 years old ) . Millennial ( 2 6 to 4 0 years old ) . Generation Z ( 2 5 years old and younger...
-
Solitary confinement, also known as restrictive housing, has been an important component of the American penitentiary system. The main criticism of solitary confinement has been that it causes...
-
On January 1, 2023, Susan Hong lent $60855 to Ben Bachu. A zero-interest-bearing note (face amount, $81000) was exchanged solely for cash; no other rights or privileges were exchanged. The note is to...
-
Engberg Company installs lawn sod in home yards. The company's most recent monthly contribution format income statement follows: Percent of Sales Variable expenses Contribution margin Fixed expenses...
-
Which decision-making tool allows one to check whether or not a course of action will actually improve the situation and is worth taking? Force field analysis Grid analysis method Pros and cons...
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
The heat of atomization is the heat required to convert a molecule in the gas phase into its constituent atoms in the gas phase. The heat of atomization is used to calculate average bond energies....
-
Draw Lewis structures for the following free radicals. (a) The ethyl radical, (b) The tert-butyl radical, (CH3)3C (c) The isopropyl radical (2-propyl radical) (d) The iodine atom CH3 CH2
-
(a) Using bond-dissociation enthalpies from Table 4-2 (page 143), calculate the heat of reaction for each step in the free-radical bromination of methane. (b) Calculate the overall heat of reaction....
-
The reaction of tert-butyl chloride with methanol Is found to follow the rate equation Rate = kf [(CH3)3C-Cl] (a) What is the kinetic order with respect to tert-butyl chloride? (b) What is the...
-
Suppose you were interested in studying the quality of conditions within a prison. What indicators would you measure to give the clearest picture of the realities of prison life? Cite the below...
-
On March 31, 2023, Panda Co. assessed its assets for impairment as part of its year-end procedures. It was found that equipment had a recoverable value of $15,000, a remaining useful life of three...
-
Petty's comparative balance sheets at December 31, 2020, and December 31, 2019, report the following (in millions). (Click the icon to view the comparative balance sheets.) Requirements Below are...

Study smarter with the SolutionInn App


