Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB, orE2: NaN3
Question:
Tell whether each of the following reactions is likely to be SN1, SN2, E1, E1cB, orE2:
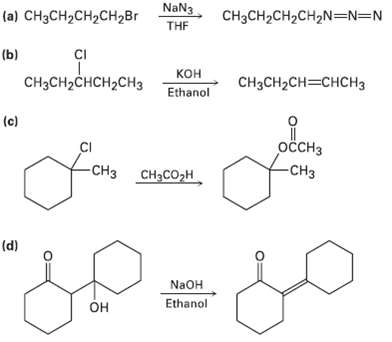
Transcribed Image Text:
NaN3 (a) CHзCH2CH2сH2Br CH3CH2CH2CH,N=N=N THE CI (b) ононалонен, кон CнзCH-CHCH2CHЗ CHзCH2CH—CHCH3 Ethanol (c) оссHз CI -CHз сн-со-н -CHз (d) NaOH Ethanol Он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
a CH3CHCHCHBr NaN3 primary The reaction occurs by an SN2 mechani...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
Tell whether each of the following reactions is an oxidation, a reduction, orneither. (a) NABH4 H20 CH;CH- CH3CH2CH2OH (b) OH 1. BH3 2. NaOH, H202
-
Tell whether each of the following reactions is an oxidation, a reduction, orneither: (a) CH3CH CH3CH2OH (b) HH + NH NCH2CH2CCH3 Br (c) 1. Mg CHCH2CHCH CCH2CH2CH3 2. H20
-
Tell whether each of the following molecules has a meso stereoisomer. (a) (b) CH CHCH CHCH Cl CH, CHCH2CH,CH CI
-
All individuals in Canada have the responsibility to take meaningful actions towards truth and reconciliation. As business professionals, we take it a step further and talk about our responsibility...
-
What some of the benefits that exist for employees who learn English
-
C4.5rules is an implementation of an indirect method for generating rules from a decision tree. RIPPER is an implementation of a direct method for generating rules directly from data. (a) Discuss the...
-
Complete a simulation of the entire process for the hydrodealkylation of toluene in Figure 6.14. Initially, let the purge/recycle ratio be 0.25 ; then, vary this ratio and determine its effect on the...
-
The optical disk project team has started gathering the information necessary to develop the project networkpredecessor activities and activity times in weeks. The results of their meeting are found...
-
Two Firefighter/Paramedics who work on opposite shifts have an ongoing personality conflict. One of the paramedics Bill Jones (a 40 year old Army veteran) has come to the supervisor on several...
-
Mr. Juan Dela Cruz is a self-employed professional based in the Philippines. He provides consulting services in the field of information technology. As a resident Filipino citizen, Mr. Dela Cruz is...
-
Which isomer would you expect to undergo E2 elimination faster, trans1-bromo-4-tert-butylcyclohexane or cis-1-bromo-4-tert-butylcyclohexane? Draw each molecule in its more stable chair conformation,...
-
Write the product you would expect from reaction of each of the following alkyl halides with (i) Na + ? SCH3 and (ii) Na + ?? OH (yellow-green = Cl): (a) (c) (b)
-
Carrington Oil produces gas 1 and gas 2 from two types of crude oil: crude 1 and crude 2. Gas 1 is allowed to contain up to 4% impurities, and gas 2 is allowed to contain up to 3% impurities. Gas 1...
-
Explain the relationship between the present value and time if discount rate is kept constant.
-
Title: Comprehensive Exploration of Accounting: Transaction, Assets, Goodwill, and Liabilities I. Introduction Accounting serves as the backbone of financial management, providing a systematic...
-
Wildhorse Company is considering two different, mutually exclusive capital expenditure proposals. Project A will cost $ 4 6 6 , 0 0 0 , has an expected useful life of 1 3 years and a salvage value of...
-
Is it appropriate for nonprofit organizations who rely upon donations to invest in risky assets such as the stock market?
-
As a tax professional, understanding the key components associated with taxation is crucial for ensuring accurate financial reporting and compliance. Accounting profit represents the profit reported...
-
Use Johnsons rule to obtain the optimum sequence for processing the jobs shown through work centers A and B. JOB TIMES (hours) Job Work Center A Work Center B a 2.50 4.20 b 3.80 1.50 C 2.20 3.00 d...
-
The MIT Sloan School of Management is one of the leading business schools in the U.S. The following table contains the tuition data for the masters program in the Sloan School of Management. a. Use...
-
Consider the reaction: A reaction mixture initially contains a CO partial pressure of 1344 torr and a H 2 O partial pressure of 1766 torr at 2000 K. Calculate the equilibrium partial pressures of...
-
Arrange each group of compounds in order of increasing acidity. (a) Phenol, ethanol, acetic acid (b) P-toluenesulfonic acid, acetic acid, chloroacetic acid (c) Benzoic acid, o-nitrobenzoic acid,...
-
What do the following pKa values tell you about the electron-withdrawing abilities of nitro, cyano, chloro, and hydroxyl groups? CH COOH CH,COOH CH COOH CHACOOH CHACOOH NO, 1.68 CN 2.46 CI 2.86 3.83...
-
Given the structure of ascorbic acid (vitamin C): (a) Is ascorbic acid a carboxylic acid? (b) Compare the acid strength of ascorbic acid (pKa = 4.71) with acetic acid. (c) Predict which proton in...
-
What are some features of Cross Laminated Timber?
-
The 201 1 price implied by PPP for the Billy bookcase in the United Kingdom was 137.49, but the actual price at that time was $29.90. What are possible reasons why the price was relatively low?
-
Within the intricate tapestry of advanced organizational ecosystems, characterized by their emphasis on continuous learning and adaptive capabilities, what strategies can organizational leaders...

Study smarter with the SolutionInn App


