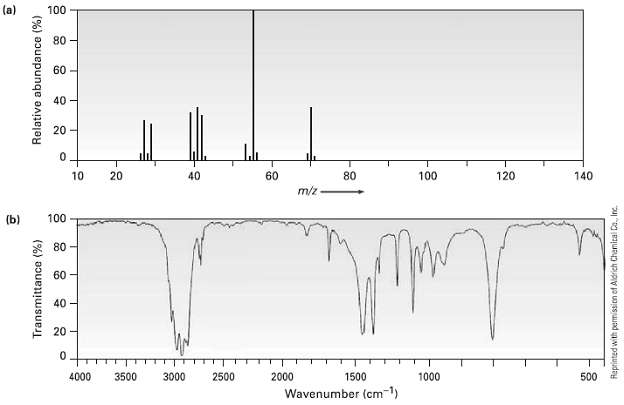
The (a) mass spectrum arid the (b) infrared spectrum of another unknown hydrocarbon is shown. Propose as
Question:
The (a) mass spectrum arid the (b) infrared spectrum of another unknown hydrocarbon is shown. Propose as many structures as youcan.
Transcribed Image Text:
(a) 100 80 60 40 20 20 60 80 100 120 140 10 40 m/z 100 (b) * 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm1) Transmittance (%) Relative abundance (%) Raprinted with permission ef Aldrich Chanical Co., Inc.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
The molecular ion M 70 corresponds to the molecular formula C5H10 This compou...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The mass spectrum and infrared spectrum of an unknown compound are shown in Figures 13.27 and 13.28, respectively. Identify the compound. Figure 13.27 The mass spectrum for Problem 28. 100 E 80 3 60...
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
Propose reasonable fragmentation mechanisms that explain why The EI mass spectrum of benzoic acid shows major peaks at m/z = 105 and m/z = 77.
-
An amplifier has three cascaded stages of amplification, each having available power gain of 10 dB and noise figure of 3 dB. i) ii) Calculate the noise factor (ratio), and noise figure (dB). If the...
-
Why are the Cournot and Bertrand models considered static? What aspects of real world behavior might be missing in static models?
-
A pizza shop owner wishes to find the 95% confidence interval of the true mean cost of a large cheese pizza. How large should the sample be if she wishes to be accurate to within $0.15? A previous...
-
What were two of the most profound influences in the development of present-day hospitals?
-
Differentiate between a bribe and a grease payment. Give an example of each.
-
Required Information Problem 17-2A (Algo) Ratios, common-size statements, and trend percents LO P1, P2, P3 [The following information applies to the questions displayed below.)] Selected comparative...
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
The (a) mass spectrum and the (b) infrared spectrum of an unknown hydrocarbon are shown. Propose as many structures as youcan. (a) 100 80 60 40 20 10 20 40 60 80 100 120 140 m/z (b) 60 40 20 - 4000...
-
Propose structures for compounds that meet the following descriptions: (a) An optically active compound C5H10O with an IR absorption at 1730 cm1 (b) A nonoptically active compound C5H9N with an 1k...
-
Collinearity is sometimes described as a problem with the data, not the model. Rather than filling the scatterplot of X1 on X2, the data concentrate along a diagonal. For example, the following plot...
-
Management accounting literature is abundant with discussions and commentaries on the changing roles of management accountants. The advent of Johnson and Kaplan's, 'Relevance Lost,' has certainly...
-
A sum of $1250 is to be divided between two people in the ratio of 2 to 3. How much does each person receive? Please include steps
-
) On January 1, 2023, Linda Corp. changed from the average cost to the dollar value LIFO method for accounting for inventory. The change results in a decrease in ending inventory of $45,497,000 for...
-
Use the spinner to find each theoretical probability a). P (a number no more than 5) b). P (an even number) c). P (a number less than 3) 8 7 1 2 6 5 3
-
In 2022, Pluto, Inc. decided to switch from traditional LIFO to Dollar Value LIFO. Under traditional LIFO, ending inventory in 2021 (the company's first year) was $2,521,000 and COGS was $14,622,000....
-
In problem #1, we used the given sample data to estimate whether there is a difference in happiness scores based on the type of diet. Using Fisher's LSD, describe and interpret any and all...
-
Refrigerant R-12 at 30C, 0.75 MPa enters a steady flow device and exits at 30C, 100 kPa. Assume the process is isothermal and reversible. Find the change in availability of the refrigerant.
-
A gas has a Henrys law constant of 0.112 M/atm. What total volume of solution is needed to completely dissolve 1.65 L of the gas at a pressure of 725 torr and a temperature of 25 C?
-
A nitro group( - NO2) effectively stabilizes a negative charge on an adjacent carbon atom through resonance: Two of the following nitrophenols are much more acidic than phenol itself. The third...
-
Classify each reaction as an oxidation, a reduction, or nether. (a) CH3 - CH2OH (b) (c) (d) (e) (f) g) (h) (i) (j) (k) (l) CrOs pyridine CH H2CrO4 CH3CH -- CH3 H3C CH3 CH3---CH3 LiAIH TiCI...
-
Show how you would convert propan-1-ol to the following compounds using tosylate intermediate. You may use whatever additional reagents are needed. (a) 1-bromopropane (b) Propan-1-1amine,...
-
Melissa Cutt is thinking about buying some shares of EZLawn Equipment, at $36.44 per share. She expects the price of the stock to rise to $43.62 over the next 3 years. During that time she also...
-
At the beginning of the week, Taylan's restaurant had a beginning inventory of food and beverages totaling $4,000. During the week, additional food and beverages purchases totaled $3,000. At the end...
-
1. Identify the auditor's role in the internal and external auditing processes. 2. Identify one law, regulation, and statue affecting healthcare

Study smarter with the SolutionInn App


