The compound whose 1H NMR spectrums is shown has the molecular formula C3H6Br2. Propose astructure. TMS O
Question:
The compound whose 1H NMR spectrums is shown has the molecular formula C3H6Br2. Propose astructure.
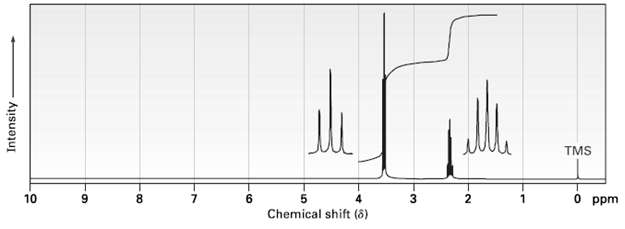
Transcribed Image Text:
TMS O ppm 10 8. 6. 1. Chemical shift (8) Intensity 6.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (21 reviews)
The unknown compound has no ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The compound whose 1 H NMR spectrum is shown has the molecular formula C 4 H 7 O 2 C1 and has an infrared absorption peak at 1740 cm ?1 . Propose a structure. TMS 10 O ppm Chemical shift (8) Intensity
-
Compound M has the molecular formula C9H12. The 1H NMR spectrum of M is given in Fig. 14.29 and the IR spectrum in Fig. 14.30. Propose a structure for M.
-
The 1H NMR spectra of three isomers with molecular formula C4H9Br are shown here. Which isomer produces which spectrum? a. b. c. (ppm) -frequency o (ppm) frequency 5 2 (ppm) frequency
-
Tom Lamont, age 30, and Lin Lamont, age 31, have been married for six years. They got married right after Tom graduated from college. They have come to you for help in planning their financial...
-
Suppose that Arnold Schwarzenegger (GAS) pays Besanko, Dranove, and Shanley (BDS) an advance of $5 million to write the script to Incomplete Contract, a movie version of their immensely popular text...
-
Three point charges are positioned in the x-y plane as follows: 5 nC at y = 5 cm, 10 nC at y = 5 cm, and 15 nC at x = 5 cm. Find the required x-y coordinates of a 20-nC fourth charge that will...
-
The University of Wisconsin at Madison completed a study titled GenderEquity Study of Faculty Pay,dated June 5, 1992. The main purpose of the study was to determine whether women are treated unfairly...
-
Moonstruck Company manufactures a reservation system with an estimated economic life of 12 years and leases it to National Airlines for a period of 10 years. The normal selling price of the equipment...
-
After watching the movie(Enron- the smartest guys in the room) and also the clip of Milton Friedman discussing Greed, what are your thoughts on the difference of "free markets" vs. regulations in...
-
A producer of inkjet printers is planning to add a new line of printers, and you have been asked to balance the process, given the following task times and precedence relationships. Assume that cycle...
-
How could you use 1H NMR, 13C NMR, and IR spectroscopy to help you distinguish between the followingstructures? "CH 3-Methyl-2-cyclohexenone 3-Cyclopentenyl methyl ketone
-
Propose structures for compounds that fit the following 1H NMR data: (a) C5H10O 0.95 (6 H, doublet, J = 7 Hz) 2.10 (3 H, singlet) 2.43 (1 H, multiplet) (b) C3H5Br 2.32 (3 H, singlet) 5.35 (1 H,...
-
Two cars traveling with the same speed move directly away from one another. One car sounds a horn whose frequency is 422 Hz and a person in the other car hears a frequency of 385 Hz. What is the...
-
Using the information obtained in the library or on the Internet, answer the following questions about Peter Mannings investment suggestions. a. What does the rating for the America West Airlines...
-
On 30 March 20X4, Jason Inc subscribed to purchase 1,600 common shares of BIT - Split Corporation for $14 per share. Jason Inc paid the subscription amount in 2 equal installments on 7 May 20X4 and...
-
1. Why are there tradeoffs in choosing a team's size and level of diversity? 2. Can you identify an example of a development project and what type of team you believed they used? Do you think this...
-
In October 2023, ABC Corp enters into negotiations with XYZ Corp to lease commercial property. ABC pays G, its outside legal counsel, $4,000 in October 2023 for services rendered by G during December...
-
Tom incurred the following expenditure in relation to his income producing property for the year ended 30 June 2023: Removal of worn linoleum flooring in the kitchen and dining room and replacing...
-
A weight loss program claims that its clients have lost, on average, more than 40 pounds over the course of a 4 -month period. A random sample of 30 participants in the weight loss program shows an...
-
Find the intercepts and then graph the line. (a) 2x - 3y = 6 (b) 10 - 5x = 2y
-
Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pressure of 1.0 atm and a mole fraction for nitrogen of 0.78.
-
Show how you would use the Williamson ether synthesis to prepare the following ethers. You may use any alcohols or phenols as your organic starting materials. (a) cyclohexyl propyl ether (b)...
-
Rank each group of compounds in order of increasing heat of hydrogenation. (a) hexa-1, 2-diene; hexa-1, 3, 5-triene; hexa-1, 3-diene; hexa-1, 4-diene; hexa-1, 5-diene; hexa-2, 4-diene. (b)
-
When N-bromosuccinimide is added to hex-1-ene in CCl4 and a sunlamp is shone on the mixture, three products result. (a) Give the structures of these three products. (b) Propose a mechanism that...
-
1) Explain how a Preschool program can help to bring a sense of belonging for the child? 2) It is important for a program to provide physical environments that are age-appropriate and that support...
-
Your company decided to organize a special dinner to appreciate all the employees and it has been proposed that the dinner to be held in the middle of December 2021. You have been appointed as the...
-
Select a two-dimensional image to analyze that is either a work of art (photograph, illustration, or painting) or an advertisement. What is the FTC analysis?Use the FTC palette to break down the...

Study smarter with the SolutionInn App


