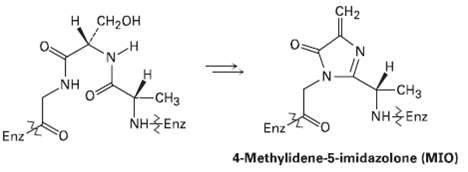
The first step in the biological degradation of histidine is formation of a 4-methylidcneirnidazol-5-one (MIO) by Cyclization
Question:
The first step in the biological degradation of histidine is formation of a 4-methylidcneirnidazol-5-one (MIO) by Cyclization of a segment of the pep- tide chain in the histidine ammonia lyase enzyme. Propose amechanism.
Transcribed Image Text:
н сн-он CH2 for -CH3 NH -СНз NHEnz NHEnz Enz Enz 4-Methylidene-5-imidazolone (MIO)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 93% (15 reviews)
Enz B Enz H CHOH HO N H CH3 NHEnz HCHOH H CH3 n...View the full answer

Answered By

Nandana Wijayarathna
I am a highly experienced writer in several areas,
Business management
Information technology
Business administration
Literature
Biology
Environmental science
History
4.50+
161+ Reviews
399+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The first step in the biological degradation of lysine is reductive amination with ?-ketoglutarate to give saccharopine. Nicotinamide adenine dinucleotide phosphate (NADPH), a relative of NADH, is...
-
The first step in the revenue recognition process is determining if a contract is in place between the seller and the customer. A contract is an agreement between two or more parties that creates...
-
The first step in the controlling process involves measuring performance. If you were John Thain, how would you measure the performance of the financial planners? Be specific.
-
You are analyzing HUE stock. You expect that the dividends over the next three years will be $1.75 in year 1, $1.90 in year 2, $2.00 in year 3 and that HUEs stock price will be $46.32. What is the...
-
Explain what the following gestures or one during a conversation. How did you reach your conclusions about each nonverbal signal? How do such signals influence your interpretation of spoken words?...
-
For the network of Fig. 2.166, sketch vo and iR. 10 V 0 Si -10 V
-
Calculate the minimum rate of work in watts for the gaseous separation at ambient conditions indicated in Figure 10.32. Figure 10.32:- Feed Separator kmol/hr Cz 30 Cz 200 nC4 370 nC5 350 nC6 50...
-
Timken Company specializes in the repair of music equipment and is owned and operated by Secilia Timken. On April 30, 2012, the end of the current year, the accountant for Timken Company prepared the...
-
Create a research paper outline about The Right to Abortion. The outline should make your points, in abbreviated form, and include your supporting evidence - i.e., cases (including the 4 legal codes...
-
Beverly Mills has decided to lease a hybrid car to save on gasoline expenses and to do her part to help keep the environment clean. The car she selected is available from only one dealer in the local...
-
Amino acids arc metabolized by a transamination reaction in which theNH2 group of the amino acid changes places with the keto group of an -keto acid. The products are a new amino acid and a new -keto...
-
Carnauba wax, used in floor and furniture polishes, contains an ester of a C32 straight-chain alcohol with a C20 straight-chain carboxylic acid. Draw its structure.
-
In 2011, an 1880-O Morgan silver dollar sold for $13,113. What was the rate of return on this investment?
-
Consider the irrigation system shown below. The system consists of a reservoir with a maximum depth of h=2.5m, 5 sections of piping with a diameter of 5cm, 2 pipe bends (r/d=3), a wide-open globe...
-
The World Economic Forum (WEF) made economic headlines with a report it published in late 2020. In the report, they predicted that 85 million jobs in the manufacturing sector worldwide would be lost...
-
R-134a flows steadily through a compressor. The inlet of the compressor is maintained at 400 kPa and the refrigerant enters the compressor superheated by 1.09C (i.e., 1.09C above the saturation...
-
REFER TO THE GIVEN PROBLEM BELOW; Country of Jurisdiction: Philippines 1. How much is the business tax if the business is Videoke Bar? 2. How much is the business tax if the business is professional?...
-
2. From the following balances of Amal, Bimal as on 31.12.2016, prepare Trading Account, Profit and Loss Account for the year ended 31.12.2016 and the Balance Sheet as at 31.12.2016. Opening stock...
-
David and Tami deposit \($150\) into their joint account at the end of each month. If their account earns 6 percent per year compounded quarterly, how long will it take them to have a total of...
-
Find the equations of the ellipses satisfying the given conditions. The center of each is at the origin. Passes through (2, 2) and (1, 4)
-
Find the pH of each mixture of acids. a. 0.075 M in HNO 3 and 0.175 M in HC 7 H 5 O 2 b. 0.020 M in HBr and 0.015 M in HClO 4 c. 0.095 M in HF and 0.225 M in HC 6 H 5 O d. 0.100 M in formic acid and...
-
How could you use IR spectroscopy to distinguish between the following pairs of isomers? a. b. c. (CH3CH2)3N and (CH3CH2CH2)2NH 0 CH.CCH,CH, and CH CHCH-CH, CH3 CHCHO and CH CHOCH
-
You are oxidizing 1-octanol with PCC to obtain octanal (eq. 7.38). How could you use IR spectroscopy to tell that the reaction was complete and that the product was free of starting material?
-
In Chapter 9 ("A Word About . . . Tautomerism and Photochromism," page 274), we discussed the difference in color between the phenol-imine (yellow) and the keto-enamine (red). Rationalize the...
-
A couple obtained a $20,000 mortgage loan at an interest rate of 10.5% compounded monthly. (Original principal equals to PV of all payments discounted at the interest rate on the loan contract) (1)...
-
What strategies and tactics are employed to manage strategic risks and uncertainties, including geopolitical instability, supply chain disruptions, and emerging competitive threats, while preserving...
-
How do strategic planners integrate ethical considerations and sustainability imperatives into strategic planning processes, balancing short-term financial objectives with long-term societal and...

Study smarter with the SolutionInn App


