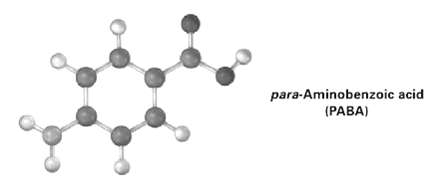
The following molecular model is a representation of para-aminobenzoic acid (PABA), the active ingredient in many sunscreens.
Question:
The following molecular model is a representation of para-aminobenzoic acid (PABA), the active ingredient in many sunscreens. Indicate the positions of the multiple bonds. And draw a skeletal structure (gray = C, red = O, blue = N, ivory =H).
Transcribed Image Text:
para-Aminobenzoic acid (PABA)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
HN Vis...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following ball-and-stick molecular model is a representation of thalidomide, a drug that causes birth defects when taken by expectant mothers (red = 0, gray = C. blue = N, ivory = H). (a) What is...
-
The following model is a representation of asparatame, C14H18N2O5, known commercially as NutraSweet. Only the connection between atoms is shown; multiple bonds are not indicated. Complete the...
-
The following molecular model is that o a carbocation. Draw two resonance structures for the carbocation, indicating the positions of the doublebonds.
-
How can a layer fold and boudinage (extend) at the same time?
-
Osprey Corporation, an accrual basis taxpayer, had taxable income for 2016 and paid $40,000 on its estimated state income tax for the year. During 2016, the company received a $4,000 refund upon...
-
When the TSL instruction is used for multiprocessor synchronization, the cache block containing the mutex will get shuttled back and forth between the CPU holding the lock and the CPU requesting it...
-
What is the January solar load fraction for a water-heating system in Washington, DC, using \(100 \mathrm{~m}^{2}\) of solar collector if the water demand is \(4 \mathrm{~m}^{3} /\) day at...
-
On January 1, 2014, Pinnacle Corporation exchanged $3,200,000 cash for 100 percent of the outstanding voting stock of Strata Corporation. On the acquisition date, Strata had the following balance...
-
If you had to speak in front of the Detroit City Council about this issue, who would you be supporting - the Hagermans or Judy Kelley? Explain your answer. People Are Making Big Money Kicking Detroit...
-
Keeper Corporations income statement for the year ended June 30, 2014, and its comparative balance sheets for June 30, 2014 and 2013 follow. Keeper Corporation Income Statement For the Year Ended...
-
Propose skeletal structures for compounds that satisfy the following molecular formulas. There is more than one possibility in each case. (a) C5H12 (b) C2H7N (c) C3H6O (d) C4H9Cl
-
Convert each of the following molecular models into skeletal structure, and give the formula of each. Only the connections between atoms are shown; multiple bonds are not indicated (gray = C, red ?...
-
If $P is invested at 100r% compounded annually, the amount A present after n years forms a geometric sequence with common ratio 1 + r. Write a formula for the amount present after n years.
-
A ramp used in lab 3: the height of the block is measured to be 10.25 cm, the distance between the support of the ramp is measuted to be 84.88 cm, whtat is the theoretical acceleration of the cart on...
-
All pizza slices at Pizza Party Zone have the same mass. Tim buys a pizza there that has a total mass of 800 grams. The pizza has 8 slices. What is the mass of one of the pizza slices on Tim's pizza
-
Explore the implications of Newton's third law of motion in terms of safety measures in activities like car crashes or sports. How can an understanding of action and reaction forces help in designing...
-
The mechanical power produced by the motor is Pmech = T. The electrical power taken by the motor from the circuit, is Pelec = Vei. What should be the theoretical relationship between Kt and Ke for...
-
An airplane performing a trick flies directly upwards at 80.7 m/s, then cuts their engine and freefalls until they are 171.1 m below their initial starting point. Determine the plane's final velocity
-
How would you expect the variation to differ between times in the Olympic marathon and times in the New York City marathon? Explain.
-
The following exercises are not grouped by type. Solve each equation. x610x -9
-
Show that the criterion for chemical equilibrium developed in the text, for a closed system at constant temperature and pressure, is also the equilibrium condition to be satisfied for closed systems...
-
Determine whether these structures represent the same compound or isomers; a) H-C. -C -C H-C-H H H b) H-C-C. H-C- H H H H-C-H H H TI C_CH C H H H c) H=C=C d) H-C-C H H-CH H-C- ...
-
Calculate the DU for these formulas and draw two constitutional isomer for each: (a) C 10 H 22 (b) C 9 H 16 (c) C 6 H 6
-
Convert these structures to skeletal structures: T a) H-C-C H H H-C-H C c) CH3CHCCHCH(CH3)2 C=C=C-H b) H H-C-H H H CH3 d) CH,CHCH,NHCH,CH CH,
-
The Village of Seaside Pines prepared the following enterprise fund Trial Balance as of December 31, 2020, the last day of its fiscal year. The enterprise fund was established this year through a...
-
In the context of digital transformation and the growing influence of artificial intelligence, what ethical considerations should leaders take into account to ensure that technology is deployed in...
-
How can ethical leadership influence corporate governance structures, and what governance practices or oversight mechanisms can be established to ensure that ethical principles are embedded in...

Study smarter with the SolutionInn App


