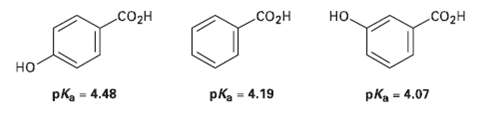
The following pKa values have been measured. Explain why a hydroxyl group in the para position decreases
Question:
The following pKa values have been measured. Explain why a hydroxyl group in the para position decreases the acidity while a hydroxyl group in the Meta position increases the acidity.
Transcribed Image Text:
но. CO2H CO2H CO2H но pka = 4.19 pKa = 4.07 %3D pka - 4.48
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
As we have seen throughout this book the influence of sub...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What do the following pKa values tell you about the electron-withdrawing abilities of nitro, cyano, chloro, and hydroxyl groups? CH COOH CH,COOH CH COOH CHACOOH CHACOOH NO, 1.68 CN 2.46 CI 2.86 3.83...
-
Explain why owner's equity increases by revenues and decreases by expenses.
-
Explain why operating leverage decreases as a company increases sales and shifts away from the break-even point.
-
Two moles of an ideal monatomic gas go through the cycle abc. For the complete cycle, 800 J of heat flows out of the gas. Process ab is at constant pressure, and process bc is at constant volume....
-
Suppose you are a team leader and one of your team members has a strong work ethic, based on his or her cultural values. Is it fair to assign this member much more work just because he or she is...
-
Derive an expression for the temperature distribution in a plane wall in which distributed heat sources vary according to the linear relation where w is a constant and equal to the heat generated per...
-
The velocity spectrum below is due to EDM damage. There are multiple sidebands in the spectrum that are usually separated by what frequency? D+ 300 R$ Pask 151 100 10202011201 Line
-
McKnight manufactures coffee mugs that it sells to other companies for customizing with their own logos. McKnight prepares flexible budgets and uses a standard cost system to control manufacturing...
-
A 8 . 3 2 m deep aquiarium has a glass observation window. The window is 2 . 5 6 m high by 4 . 7 m wide. If the bottom of the window is located 3 . 4 m above the bottom of the aquarium, how much...
-
Sanderson is a bus driver who earns a monthly gross income of $3,500. His wife Norma is the manager of an automotive parts store and she earns gross income of $2,250 every two weeks. The couple has...
-
How would you carry out the following transformations? More than one step is required in each case. (a) H (b) Ph "Co "
-
3-Methyl-2-hexenoic acid (mixture of E and Z isomers) has been identified as the substance responsible for the odor of human sweat. Synthesize the compound from starting materials having five or...
-
a. Describe the recording and control function of financial accounting. b. Explain the role of financial accounting with regard to the presentation of final financial statements.
-
PRC stand for and in what function is this collected?
-
A 1 2 . 0 m lenghh of copper wore has a resistance of 1 . 5 0 ohms. How long must an aluminium wire with the same cross sectional area be to have the same resistance? ( resistivities of copper and...
-
What does "open interest" in an option quote indicate?
-
Max Smith is a tax preparer in 2023 who fails to sign a return for two of his clients on the same day. What amount will he be penalized?
-
Originally, what was one of the larger regular costs of operating a McDonald franchises?
-
True or False: When dealing with multiple alternatives having unequal lives, the planning horizon equals the least common multiple of lives.
-
(a) What do data breach notification laws require? (b) Why has this caused companies to think more about security?
-
Which solid has the highest melting point? Why? Ar(s), CCl 4 (s), LiCl(s), CH 3 OH(s)
-
Give the electronic configurations of each of the following atoms and ions. Identify the valence electrons and valence orbitals in each. (a) Chloride ion, Cl- (b) Sodium atom
-
Draw an orbital interaction diagram corresponding to Fig. 1.14 for each of the following species. Indicate which are likely to exist as diatomic species, and which would dissociate into monatomic...
-
When two different species have the same number of electrons, they are said to be isoelectonic. Name the species that satisfies each of the following criteria: (a) The singly charged positive ion...
-
Using the following information: a. The bank statement balance is $3,048. b. The cash account balance is $3,300. c. Outstanding checks amount to $755. d. Deposits in transit are $809. e. The bank...
-
Determine the average tax rate and the marginal tax rate for each of the following instances: Use the Tax Tables for taxpayers with taxable income under $ 1 0 0 , 0 0 0 and the Tax Rate Schedules for...
-
A machine was bought on 1 st January 2 0 1 7 for $ 1 2 , 0 0 0 . The policy is to depreciate the machine at 1 0 % on reducing balance method at the end of each year, ending on 3 1 st December....

Study smarter with the SolutionInn App


