How would you carry out the following transformations? More than one step is required in each case.
Question:
How would you carry out the following transformations? More than one step is required in each case.
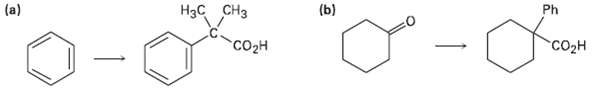
Transcribed Image Text:
(a) Нас сHз (b) Ph "Coн "Соон
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (20 reviews)
CH3CHCICH3 AICI 3 H3 CH3 H3 CH3 1 PhMgBr ether 2 HO COH C6H5 H ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
How would you carry out the following transformations? "Co CH2 C Lc
-
How would you carry out the following conversions? More than one step is needed in eachcase. "
-
A monatomic ideal gas that is initially at a pressure of 1.50 X 105 Pa and has a volume of 0.0800 m' is compressed adiabatically to a volume of 0.0400 m3. (a) What is the final pressure? (b) How much...
-
What can you do this week to help prepare yourself to become a multicultural leader?
-
Sydney invests $100 every month into an account that pays 5% annual interest, compounded monthly. Benny invests $80 every month into an account that pays 8% annual interest rate, com- pounded...
-
What is a System Element?
-
Listed below are five procedures followed by Gilmore Company. 1. Employees are required to take vacations. 2. Any member of the sales department can approve credit sales. 3. Paul Jaggard ships goods...
-
26. Consider the AVL tree resulting from sequentially inserting the following numbers into an empty AVL tree: 40, 60, 55, 15, 20, 2, 25, 30. What is the sum of the numbers in the leaf nodes in the...
-
The uniform plate of mass m is released from rest while in the position shown. Determine the initial angular acceleration a of the plate and the magnitude of the force supported by the pin at O. The...
-
The pK a ?s of five p-substituted benzoic acids (YC 6 H 4 CO 2 H) follow. Rank the corresponding substituted benzenes (YC 6 H 5 ) in order of their increasing reactivity toward electrophilic aromatic...
-
The following pKa values have been measured. Explain why a hydroxyl group in the para position decreases the acidity while a hydroxyl group in the Meta position increases the acidity. . CO2H CO2H...
-
A sales-based ranking of software companies provided by Yahoo! Finance on November 5, 2008, places Oracle Corporation third behind sales leaders Microsoft Corporation and IBM Software. Typical of...
-
Caltex Ltd's cost of equity is 15 percent, and its "after-tax" cost of debt is 5 percent. If the company is financed with 40 percent ordinary shares, what is the weighted average cost of capital for...
-
What is the process used when replacing a fund manager. What are the issues that need to be considered in the process? Discuss the considerations and problems in replacing a manager and the costs...
-
How do interdisciplinary approaches, incorporating insights from sociology, anthropology, and economics, enrich our comprehension of empowerment processes ?
-
what extent do postcolonial theories inform our understanding of empowerment dynamics in global contexts ? Explain
-
How might advancements in technology and globalization reshape the landscape of empowerment, and what ethical considerations arise in the pursuit of digital empowerment initiatives ?
-
How are surface processes different from tectonic processes?
-
A horizontal annulus with inside and outside diameters of 8 and 10 cm, respectively, contains liquid water. The inside and outside surfaces are maintained at 40 and 20oC, respectively. Calculate the...
-
Which solid has the highest melting point? Why? C(s, diamond), Kr(s), NaCl(s), H 2 O(s)
-
The compound benzenc has only one type of carbon-carbon bond, and this bond has a length intermediate between that of a single bond and a double bond. Draw a resonance structure of benzene that,...
-
The compound benzenc has only one type of carbon-carbon bond, and this bond has a length intermediate between that of a single bond and a double bond. Draw a resonance structure of benzene that,...
-
Give the electronic configurations of each of the following atoms and ions. Identify the valence electrons and valence orbitals in each. (a) Chloride ion, Cl- (b) Sodium atom
-
1. Explain what the "Deinstitutionalization Mandate" is. This should be done in at least two paragraphs. 2. In another two paragraphs, discuss how status offenses are connected to the...
-
You are the owner of a very small business that sells gourmet coffee. You sell only one product, a 12-ounce bag of whole-bean French roast coffee. You sell each bag of coffee for $14 each, but due to...
-
Find the instantaneous rate of change of the function f(t)=3+5t when t=-3.

Study smarter with the SolutionInn App


