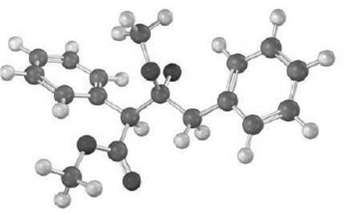
The following structure represents an intermediate formed by addition of ester emulation to a second ester molecule.
Question:
The following structure represents an intermediate formed by addition of ester emulation to a second ester molecule. Identify the reactant, the leaving group, and theproduct.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
The enolate of methyl phenylacetate adds to a second molec...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following structure represents tetrahedral alkoxide ion intermediate formed by addition of a nucleophile to a carboxylic acid derivative. Identify the nucleophile, the leaving group, the starting...
-
The following structure represents a tetrahedral alkoxide-ion intermediate formed by addition of a nucleophile to a carboxylic acid derivative. Identify the nucleophile, the leaving group, the...
-
The following structure represents the carbocation intermediate formed in the addition reaction of HBr to two different alkenes. Draw the structures ofboth.
-
Misty Cumbie worked as a waitress at the Vita Caf in Portland, Oregon. The caf was owned and operated by Woody Woo, Inc. Woody Woo paid its servers an hourly wage that was higher than the states...
-
What types of structures would a firm consider if it were greatly expanding in global operations? What types of organizing problems would it be most likely to encounter?
-
A resistance array is connected to a load resistor R and a 9-V battery as shown in Fig. 4.151. (e) Find the value of R such that vo = 1.8 V. (f) Calculate the value of R that will draw the maximum...
-
A flat plate of width \(1 \mathrm{~m}\) and length \(0.2 \mathrm{~m}\) is maintained at a temperature of \(32^{\circ} \mathrm{C}\). Ambient fluid at \(22^{\circ} \mathrm{C}\) flows across the top of...
-
During the month of February, Morrisey Corporations employees earned wages of $74,000. Withholdings related to these wages were $5,661 for Social Security (FICA), $7,100 for federal income tax, and...
-
10. From the following information prepare marginal costing statement and calculate BEP and PVR Fixed cost Ro 120000 Variable cost RO 200000 Sales RO 1000000 Direct wages RO 150000 Direct materials...
-
Create a PowerPoint presentation on the critical points in philosophy for the Existence of God (thesis) and the Problem of Evil (antithesis). The key point of the presentation is a collective group...
-
What ketone or aldehydes might the following enone have been prepared from by aldolreaction? (b) (a)
-
The following molecule was founded by an intermolecular aldol reaction. What dicarbonyl precursor was used for itspreparation?
-
What is the margin of safety (MOS), and for what is it used?
-
Secondary Analysis: This week visit the Keizer Online Library and find a full-text article on secondary analysis in research. Find a full-text article, download it, highlight it, and upload it to the...
-
Thornton Electronics currently produces the shipping containers it uses to deliver the electronics products it sells. The monthly cost of producing 9,300 containers follows. Unit-level materials...
-
Solve the following Problem 6-6 Jerry wants to withdraw $2,000 per year for the next three years starting in one year. If the bank pays interest annually at 3%, how much must he put in today so...
-
Question 1: Identify and describe the core group performance factors Question 2: Describe the difference between formal and informal groups and give a specific example of each. Describe...
-
St. Johns Paul College of Engineering received the following fees from the students. Tuition fees $10,000 Library fees $500 Special fees $1,000 The college paid $2,000 in examination fees to the...
-
Does social responsibility mean something different to your employee behavior than your behavior as a manager? Do you think a business can be socially responsible when its employees and managers are...
-
Havel says the grocer doesnt believe what is on the sign and indeed, he says the grocers customers will barely notice it. But Havel maintains that the sign serves a specific function. How would you...
-
Write Lewis structures for each molecule or ion. Use expanded octets as necessary. a. C. CIFS Cl3PO b. AsF6 d. IFs
-
A solution of pure (S)-2-iodobutane ([a] = + 15.90( in acetone is allowed to react with radioactive iodide, 131I , until 1.0% of the iodobutane contains radioactive iodine. The specific rotation of...
-
(a) Optically active 2-bromobutane undergoes racemization on treatment with a solution of KBr. Give a mechanism for this racemization. (b) In contrast, optically active butan-2-ol does not racemize...
-
For each pair of compounds, predict which compound has the higher boiling point. Check Table 6-2 to see if your prediction was right, then explain why that compound has the higher boiling point. (a)...
-
Joey set up a lawn-mowing business in his neighborhood. He currently has 7 customers that want their lawns mowed each week, for which he charges $25.00 each. Joey spends $10.00 a week in gas and...
-
Vanessa has an outstanding bond with a 7.10 % semi-annual coupon and has 16 years left to mature. The current market rate is 10.50% per annum. What is the current yield of Vanessa's Bond.
-
Estimate the hydrogen consumption required to completely remove the sulfur from a hydrotreater feedstock and to reduce the nitrogen content of the product to 15 ppm by weight. The 48.5 API naphtha...

Study smarter with the SolutionInn App


