The following tertiary alkyl bromide does not undergo a nucleophilic substitution reaction by either SN1 or SN2
Question:
The following tertiary alkyl bromide does not undergo a nucleophilic substitution reaction by either SN1 or SN2 mechanisms.Explain.
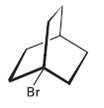
Transcribed Image Text:
Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
The first step in an Syl displacement is dissociati...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following tertiary alkyl bromides undergo an SN1 reaction in aqueous acetone to form the corresponding tertiary alcohols. List the alkyl bromides in order of decreasing reactivity. Br CH CCH3...
-
Show how you might use a nucleophilic substitution reaction of 1-bromopropane to synthesize each of the following compounds. (You may use any other compounds that are necessary.) (a) (b)...
-
Furfuryl chloride can undergo substitution by both SN2 and SN1 mechanisms. Since it is a 1° alkyl halide, we expect SN2 but not SN1 reactions. Draw a mechanism for the SN1 reaction shown below,...
-
Complete your review of the Stevens District Hospital Strategic Planning Scenario, you have been asked to provide a presentation to the governing board of the hospital. This board is comprised of the...
-
Although the board of directors is responsible for filling the CEO position, how could HR managers support the board with a succession management program?
-
Sketch a curve similar to that in Fig. 4.8 showing in general terms the energy changes that arise from rotation about the C2-C3 bond of 2-methylbutane. You need not concern yourself with the actual...
-
Burger Buddy Restaurants grants area franchises to operate up to two restaurants within a 100 square mile area. The Uniform Franchise Offering Circular reveals that, following payment of the lump sum...
-
Van Emburgh Technology has two divisions, Consumer and Commercial, and two corporate service departments, Tech Support and Accounts Payable. The corporate expenses for the year ended December 31,...
-
If total energy absorbed must equal total energy released, how is it possible for an atom to absorb visible light and release heat? Explain.
-
Refer to Exercise 3.95. Given that a student, selected at random, is found to have an extensive understanding of physics, what is the probability that the student has (a) an extensive understanding...
-
In light of your answer to problem 11.42, what product might you expect from treatment of 4-hrorno-l-butanol withbase? Base BrCH2CH2CH2CH2
-
In addition to not undergoing substitution reactions, the alkyl bromide shown in Problem 11 .45 also fails to undergo an elimination reaction when treated with base. Explain.
-
Use the appropriate limit laws and theorems to determine the limit of the sequence or show that it diverges. c n = 9n/n!
-
12. Solve the given linear congruence. Show all necessary work. A) 2x+5=3 mod(7) B) 5x-3=8 mod(7) C) 3x+1=x-3 mod(7)
-
4. Given the following sorted array: a. [2 points] Use Binary Search to determine how many and what comparisons are required to search for 101. b. [2] points Use Binary Search to determine how many...
-
Direct labor cost $10,500 Purchases of raw materials 16,500 Factory supervisor salary 2,250 Office Supplies used 675 Factory Insurance 1,350 Commissions paid Commissions paid 2,500 Advertising 1,800...
-
Explain the concept of "lazy loading" in memory management. How does it relate to demand paging, and what are the benefits and potential drawbacks of this approach ?
-
The capital accounts of Hassan Khan and Dmitri Palovich have balances of $76,000 and $54,000, respectively, on January 1, 20Y4, the beginning of the fiscal year. On July 10, Khan invested an...
-
Energy in the sun is produced by the fusion of four protons into a helium nucleus. The process involves several steps, but the net reaction is simply \(4 \mathrm{p} ightarrow{ }^{4} \mathrm{He}+\)...
-
Digital Fruit is financed solely by common stock and has outstanding 25 million shares with a market price of $10 a share. It now announces that it intends to issue $160 million of debt and to use...
-
Calculate K p for each reaction. a. NO4(8) = 2 NO(g) b. N(g) + 3 H(g) = 2 NH3(g) c. N(g) + O(g) 2 NO(g) K = 5.9 x 10- (at 298 K) K = 3.7 x 108 (at 298 K) K = 4.10 x 10-1 (at 298 K)
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Predict the products of the following reactions. (a) Cyclopentyl methyl ketone + excess Cl2 + excess NaOH (b) 1-cyclopentylethanol + excess I2 + excess NaOH (c) Propiophenone + excess Br2 + excess...
-
Which compounds will give positive iodoform tests? (a) 1-phenylethanol (b) Pentan-2-one (c) Pentan-2-ol (d) Pentan-3-one (e) Acetone (f) Isopropyl alcohol
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


