In light of your answer to problem 11.42, what product might you expect from treatment of 4-hrorno-l-butanol
Question:
In light of your answer to problem 11.42, what product might you expect from treatment of 4-hrorno-l-butanol withbase?
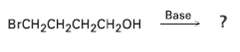
Transcribed Image Text:
Base BrCH2CH2CH2CH2он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Methoxide removes a proton from the hydrox...View the full answer

Answered By

Sagar Kumar
I am Mechanical Engineer with CGPA of 3.98 out of 4.00 from Pakistan. I went to Government Boys Degree College, Sehwan for high school studies.
I appeared in NUST Entrance Exam for admission in university and ranked #516. My mathematics are excellent and I have participated in many math competitions and also won many of them. Recently, I participated in International Youth Math Challenge and was awarded with Gold Honor. Now, I am also an ambassador at International Youth Math Challenge,
I have been teaching when I was in 9th class class year 2012. I have taught students from 6th class to university level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In light of your answer to Problem 11.61, explain why one of the following isomers undergoes E2 reaction approximately 100 times as fast as the other. Which isomer is more reactive, andwhy? (a) CI...
-
In light of your answer to Problem 4.44, draw the two chair conformations of 1, 1, 3-trimcthylcyclohexanc, and estimate the amount of strain energy in each. Which conformation is favored?
-
In light of your answer to Problem 11.49, which alkene, F or Z, would you expect from an E2 reaction on the tosylate of (2R, 3R)-3-phenyl-2-butanol? Which alkene would result from E2 reaction on the...
-
With reference to the Auditors Report, answer the following questions: The recent final audit report of an FMCG carries a line which is read as in the manner so required and respectively give a true...
-
Based on the information given, what developmental approaches were part of McDonald's career development? What approaches would you recommend for preparing P&G's next top executives?
-
Using the pKa values of analogous compounds in Table 3.1, predict which would be the stronger base. (a) (b) (c) (d) or (CHa),Cor O: HO or
-
Earnings manipulation has been the subject of considerable attention both in the financial press and in academic research. For example. The Wall Street Journal, Forbes Magazine, and Business Week...
-
Absorption versus variable costing Grunewald Company manufacturers a professional grade vacuum cleaner and began operations in 2011. For 2011, Grunewald budgeted to produce and sell 20,000 units. The...
-
The purpose of one-way ANOVA is to compare multiple means from the variations of the mean. The null hypothesis is Ho = 2 = = k and alternate is HA i Hj. We have the sample data in the table: Y1 Y2...
-
We set the potential energy in the particle in the box equal to zero and justified it by saying that there is no absolute scale for potential energy. Is this also true for kinetic energy?
-
Show the stereochemistry of the epoxide you would obtain by formation of a bromohydrin from trans-2-butene, followed by treatment with base.
-
The following tertiary alkyl bromide does not undergo a nucleophilic substitution reaction by either SN1 or SN2 mechanisms.Explain. Br
-
Instant Clothing Alterations is run by Marc Giannopolous in the local shopping centre. The business prepared the following unadjusted and adjusted trial balances at 30 June 2016: INSTANT CLOTHING...
-
What is the status of the legal personhood held by women who have experienced coerced sterilization with specific case references in Canada? You will need to research this very topic based on...
-
Discuss the role of an ombudsman/ombudsperson and how it relates and differentiates from the role of Internal Audit. Do companies with an internal audit function require an ombudsman/ombudsperson? Do...
-
1. Briefly outline how cases get the Supreme Court. 2. What are the arguments for and against judicial review? 3. What were the Alien and Sedition Laws? Are the issues surrounding them comparable to...
-
Coma Corporation v. Kansas Department of Labor Who is the plaintiff? The defendant? The appellant? The appellee? What is the history of the case? Possible questions to answer would be: Who won at...
-
Find the loop invariant, variant and weakest pre-condition in the following function. Prove total correctness of this function. What do you think this function is computing. int funl (int x) { int y...
-
A lamp flashes at the origin. Then, \(1 \mu\) s later, a lamp flashes at a point \(150 \mathrm{~m}\) from the origin. How fast, as a fraction of \(c\), would an astronaut need to travel so that he...
-
Determine the volume of the parallelepiped of Fig. 3.25 when (a) P = 4i 3j + 2k, Q = 2i 5j + k, and S = 7i + j k, (b) P = 5i j + 6k, Q = 2i + 3j + k, and S = 3i 2j + 4k. P
-
Write an equilibrium expression for each chemical equation involving one or more solid or liquid reactants or products. a. CO3- (aq) + HO(1) b. 2 KCIO3(s) = 2 KCl(s) + 3 O(g) c. HF(aq) + HO(1) H3O+...
-
Phenylacetone can form two different enols. (a) Show the structures of these enols. (b) Predict which enol will be present in the larger concentration at equilibrium. (c) Propose mechanisms for the...
-
An enolate is a very strong nucleophile. Bromine is a strong electrophile, so it can react with much weaker nucleophiles. Give mechanisms for the reactions of bromine with cyclopentene and with...
-
Propose a mechanism to show how acetophenone undergoes base-promoted chlorination to give trichloroacetophenone.
-
In the context of solid-liquid extraction, how do particle size distribution, porosity, and surface area of the solid matrix impact extraction kinetics and overall efficiency? Provide examples of...
-
Discuss the challenges and opportunities associated with the extraction of bioactive compounds from natural sources using environmentally benign solvents and green extraction techniques, such as...
-
The driver of a car traveling at 31.9 m/s applies the brakes and undergoes a constant deceleration of 1.12 m/s 2 . How many revolutions does each tire make before the car comes to a stop, assuming...

Study smarter with the SolutionInn App


