The oxymercuration reaction can be run in a methanol as the solvent rather than water. Predict the
Question:
The oxymercuration reaction can be run in a methanol as the solvent rather than water. Predict the product of this reaction.
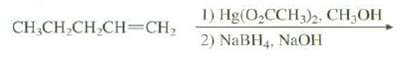
Transcribed Image Text:
1) Hg(O,CCH)2, CH3OH 2) NaBH4, NAOH CH,CH,CH CH=CH;
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
In the oxymercuration reaction the mercury electrophile adds to the double bond ...View the full answer

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What kinds of methods can be run in a C# thread?
-
Methanol (CH3OH) can be made by the reaction of CO with H2: (a) Use thermochemical data in Appendix C to calculate (Ho for this reaction. (b) To maximize the equilibrium yield of methanol, would you...
-
The following reaction does not produce the product shown. (a) Predict the major product from the conditions shown above, and write a detailed mechanism for its formation. (b) What reaction...
-
In a LVDT, the two secondary voltages a. Are independent of the core position b. Are always in phase quadrature Oc. Vary equally depending on the core position O d. Vary unequally depending on the...
-
What is descriptive analytics? What are the various tools that are employed in descriptive analytics?
-
The air in your room is composed mostly of oxygen (O2) and nitrogen (N2) molecules. The oxygen molecules are more massive than the nitrogen molecules. (a) Is the rms speed of the O2 molecules greater...
-
Consider an asset whose price follows the geometric Brownian motion process where \(z\) is a standard Wiener process. (a) At time \(t\) (when \(S(t)\) is known), what is the expected value of the...
-
Willow Creek Nursery, with an authorization of 75,000 shares of preferred stock and 200,000 shares of common stock, completed several transactions involving its stock on October 1, the first day of...
-
The number of customers arriving at Soda Mart follows a Poisson distribution with a mean of 22 customers per hour. What is the probability 40 customers will arrive in two (2) hours?
-
Your brother has just started a new job as the Controller of an IESBA restricted audit client. You do not serve on the audit engagement. What steps must you take to ensure your independence is not...
-
Show the steps in the mechanism and predict the product that would be formed in thisreaction. Br2 CH,
-
The tautomerization of an enol to a ketone is catalyzed by either acid or base. In the acid-catalyzed mechanism H+ is added in the first step (see Figure). In the base-catalyzed mechanism, H+ is...
-
1. Visit the company website (www.oprah.com/index.html) and identify and describe the different product lines that it markets? 2. How would you describe its product line breadth? 3. Review the...
-
A sounding rocket, launched vertically upward with an initial speed of 74.0 m/s, accelerates away from the launch pad at 6.00 m/s. The rocket exhausts its fuel, and its engine shuts down at an...
-
3. Show that the lateral displacement s of a ray of light penetrating a rectangular glass plate of thickness t is given by: S t sin(0-0) Cos 0
-
A 9-V battery is used to charge a 4-C capacitor through a resistor of 10052. Find (a) The initial current through the circuit. (b) The final charge on the capacitor after completely being charged....
-
Two uniform thin rods each of length L. One of mass m lies on the x axis with one end at origin and the other of mass 2m on the yaxis with one end at origin. The y-component of the center of mass is
-
The resistor symbols on the circuit diagrams have been designed for theirequivalent color codes. One of the resistors used in each Circuit does not equate to thevalue beside it. Pleage try to figure...
-
The natural rate of unemployment varies over time, with changes in demographics, the structure of the economy, and government policies. For its goal of high employment, why would it be crucial for...
-
Find the intercepts and then graph the line. (a) 2x - 3y = 6 (b) 10 - 5x = 2y
-
At 35C, K = 1.6 10 -5 for the reaction If 2.0 moles of NO and 1.0 mole of Cl 2 are placed into a 1.0-L flask, calculate the equilibrium concentrations of all species. 2NOCI(g)2NO(g) + Cl(g)
-
Glucosamine, one of the eight essential monosaccharide?s (Section 25.7), is biosynthesized as its 6-phosphate derivative from fructose 6-phosphate by reaction with ammonia. Propose a mechanism....
-
Gentiobiose, a rare disaccharide found in saffron and gentian, is a reducing sugar and forms only n-glucose on hydrolysis with aqueous acid. Reaction of Gentiobiose with iodomethane and Ag20 yields...
-
Amygdalin, or laetrile, is a cyanogenic glycoside isolated in 1830 from almond and apricot seeds. Acidic hydrolysis of amygdalin liberates HCN, along with benzaldehyde and 2 equivalents of D-glucose....
-
Suppose the correlation between the stock euro returns of Siemens and the USD/EUR exchange rate is 0.2. The standard deviation of the USD/EUR is 10% and the standard deviation of Siemens's stock euro...
-
list and describe the three key client-related factors that the advisor is required to consider when developing a "suitable" investment portfolio for their client. Please cite resources used
-
Year 1 2 3 Amount ($) 2000 3000 4000 An investment made today will pays you the above cash flows at the end of each year. If your required rate of return is 5% annual interest, how much will you pay...

Study smarter with the SolutionInn App


